Abclon Unveils Complete Tumor Suppression Data for Prostate Cancer Bispecific Antibody 'AM109'
AACR 2026 Poster Presentation
Complete Remission Achieved with Ultra-Low Dose
Superior Efficacy Demonstrated Compared to Pfizer's Utomilumab
Abclon's prostate cancer drug candidate has demonstrated preclinical results showing complete tumor remission with an ultra-low dose. The candidate achieved complete remission in animal studies using only a minimal dose and also exhibited superior efficacy compared to a competing drug from Pfizer.
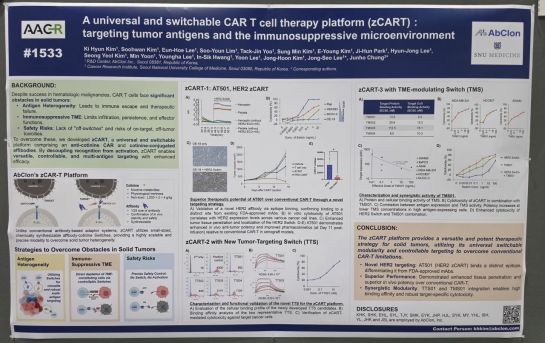
Poster of Abclon exhibited at the American Association for Cancer Research (AACR) 2026 held in San Diego, USA, from the 17th to the 22nd (local time). Abclon
View original imageAbclon announced on the 21st that it had presented preclinical data for its next-generation CAR-T platform "zCAR-T" and bispecific antibody candidate "AM109" in a poster session at the American Association for Cancer Research (AACR) 2026, held from the 17th to the 22nd (local time) in San Diego, USA.
AM109 is a next-generation immuno-oncology drug candidate developed using Abclon's proprietary bispecific antibody platform, AffiMab. It is a bispecific antibody targeting both PSMA, a protein overexpressed on the surface of prostate cancer cells, and the T cell immunostimulatory factor 4-1BB (CD137). By simultaneously engaging both targets, the drug is designed to direct immune cells to effectively attack cancer cells.
According to the newly released data, AM109 selectively and robustly activates T cells only in the presence of PSMA-expressing cancer cells within the tumor microenvironment. The animal studies demonstrated a target-dependent mechanism that minimizes side effects by not triggering an immune response in healthy tissues where cancer cells are absent. Notably, complete tumor remission was achieved with an extremely low dose of just 0.1 to 0.3 mpk. This represents a differentiated anti-cancer efficacy compared to Pfizer's utomilumab, a standard 4-1BB-targeting drug. AM109 also maintained structural and functional stability for over 12 weeks, and showed excellent pharmacokinetics and tolerability in vivo.
Also presented was zCAR-T, a next-generation solid tumor cell therapy platform independently developed by Abclon to overcome the limitations of existing CAR-T therapies. While CAR-T therapies have demonstrated remarkable efficacy in hematologic cancers, their application to solid tumors has been limited by severe toxicities such as cytokine release syndrome (CRS) and the barriers posed by the tumor microenvironment. zCAR-T addresses these challenges by finely controlling CAR-T cell activity through the adjustable dosing of a switch molecule that binds to the tumor antigen.
According to the data unveiled at the conference, the zCAR-T platform maintains potent anti-tumor efficacy comparable to traditional CAR-T therapies, while the new switch mechanism enables control not only over the tumor itself but also the tumor microenvironment. Abclon plans to expedite domestic clinical trials for AT501, one of its zCAR-T pipeline assets, and is preparing to move beyond the preclinical stage into full-scale human clinical trials.
Hot Picks Today
!["Only the Top 1% Winning Big in Stocks Smile... '300 Million Won Splurges' or '10-Won Wars,' the Middle Disappears [K-Shaped Consumption Era]①"](https://cwcontent.asiae.co.kr/asiaresize/93/2026041409371024802_1776127030.jpg) "Only the Top 1% Winning Big in Stocks Smile......
"Only the Top 1% Winning Big in Stocks Smile......
- [Exclusive] Latest Cancer Treatment Costs Rise by 40 Million Won... War Disrupts...
- "Finally Coming"... Samsung Electronics, SK hynix 2x Leveraged ETF to Be Listed ...
- Female Game Caster Makes Bold Move After Criticism Over "Short Skirt" on Broadca...
- Couple Secretly Making Love on Mountain Summit... Broadcast Live on the Internet
An Abclon representative stated, "During the conference, our business development lead is conducting partnering meetings with multiple companies onsite, and we will focus our full organizational capabilities to ensure these efforts lead to tangible outcomes such as technology licensing and joint development."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.



!["They Said It Was 'Delicious' Milk... Is the 'List of Fake Milk Products in Korea' for Real? [Matjal X-File]"](https://cwcontent.asiae.co.kr/asiaresize/307/2023072413274517286_1690172865.jpg)



