"Be Sure to Use Contraception When Taking Isotretinoin Medication"
MFDS Strengthens 'Pregnancy Prevention Program' Including Warning Label Requirements
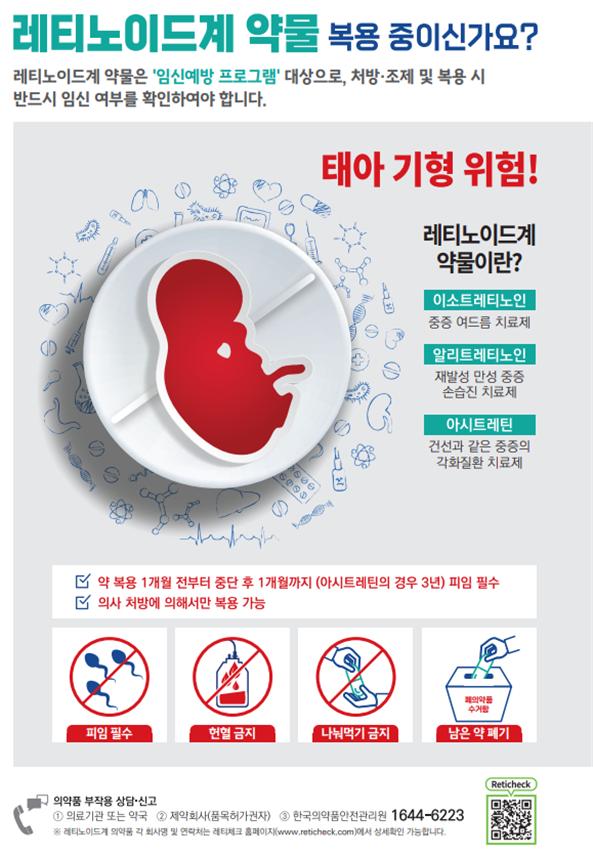
[Asia Economy Reporter Lee Gwan-ju] The Ministry of Food and Drug Safety announced on the 19th that it will strengthen the 'Pregnancy Prevention Program' by emphasizing cautionary statements on the containers and packaging of retinoid drugs, including the acne treatment ingredient 'Isotretinoin,' which poses a risk of fetal malformations when taken during pregnancy.
Retinoid drugs include Isotretinoin, Alitretinoin (for hand eczema), Acitretin (for psoriasis), and others.
The Ministry of Food and Drug Safety plans to inscribe and emphasize cautionary statements such as 'Contraception is mandatory before and after using this product for a certain period' on product containers and packaging, improve the readability of patient consent forms and leaflets, insert QR codes on products, and distribute safety use posters to hospitals and clinics. Packaging changes will be applied to products shipped from the second half of this year, considering the preparation period.
Additionally, by utilizing the 'Drug Utilization Review (DUR)' service, cooperation has been requested from the Health Insurance Review and Assessment Service to enable doctors and pharmacists to confirm pregnancy status and provide guidance on contraceptive compliance and usage precautions to all women of childbearing age when prescribing or dispensing these products.
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
Retinoid drugs are known to increase the likelihood of causing fetal malformations during pregnancy. The Ministry of Food and Drug Safety classifies them as 'Class 1' contraindicated drugs that must never be taken during pregnancy.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.