KRICT Applies for Phase 1 Clinical Trial of 'Immune-Boosting' COVID-19 Vaccine
[Asia Economy Reporter Kim Bong-su] The Korea Research Institute of Chemical Technology (KRICT) announced on the 30th that it has developed a COVID-19 vaccine candidate (IN-B009) and submitted an application for Phase 1 clinical trials to the Ministry of Food and Drug Safety.
This candidate was developed by KRICT in June last year and licensed to the private company 'inno.N'. After approval from the Ministry of Food and Drug Safety, inno.N plans to evaluate the safety and efficacy of the vaccine in healthy adults. It is a recombinant protein vaccine produced by manufacturing viral antigen proteins using genetic recombination technology and administering them.
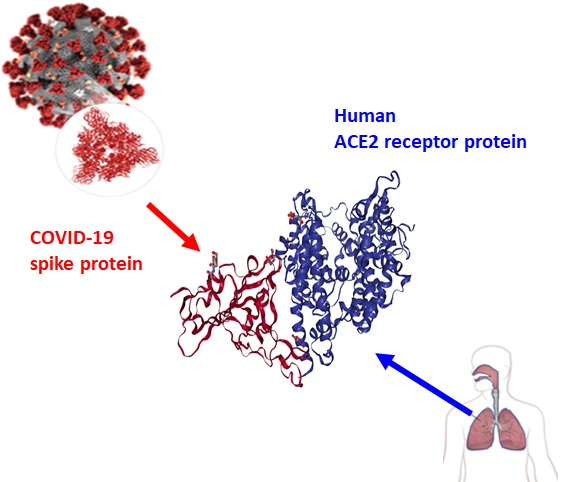
Previously, the Virus Prevention Team of the Emerging Virus Fusion Research Division at KRICT produced the spike protein segment that binds to host cells using the genetic information of the COVID-19 virus. When this protein vaccine is injected, it induces the production of antibodies, specifically neutralizing antibodies, that disable the virus from binding to receptors on human cells in the future.
The research team developed new patented technology to increase the neutralizing antibody production rate of the vaccine and enhance cellular immune activation. Safety was also confirmed through non-clinical trials using primates at the Safety Evaluation Research Institute. Based on experimental data proving excellent efficacy and safety, KRICT and inno.N are preparing to publish a paper. The vaccine also demonstrated excellent efficacy against various variant viruses such as those from the UK and South Africa.
KRICT plans to continue developing new vaccine candidates for COVID-19 variant strains and emerging and re-emerging viral infectious diseases by utilizing the 'new platform technology for high-efficacy vaccine development' along with research support necessary for COVID-19 vaccine clinical trials.
Hot Picks Today
!["Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]](https://cwcontent.asiae.co.kr/asiaresize/93/2025061015355092669_1749537351.jpg) "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
"Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- "Can I Buy a House With This Salary?"... ChatGPT Offers Instant Financial Insights When You Link Your Accounts
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
Lee Mi-hye, President of KRICT, stated, "We hope to contribute to public health and safety through the localization of a new COVID-19 vaccine with excellent efficacy and safety."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.