Coreline Soft Selected for 2.2 Billion Won Pan-Ministry Project...Pursuing Global Clinical Trials and FDA Approval
Full-Scale Overseas Clinical Trials for U.S. FDA Approval
On May 7, Coreline Soft, a medical artificial intelligence (AI) company, announced that it will begin overseas clinical trials to obtain approval from the U.S. Food and Drug Administration (FDA). The project for conducting these clinical trials has been selected for the "2026 1st Pan-Ministry Advanced Medical Device R&D Project" and will receive a total of 2.2 billion won in government support over the next three years.
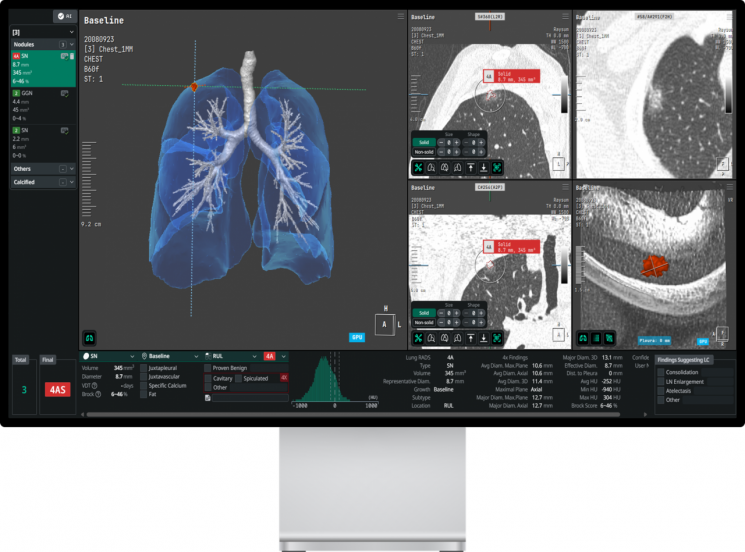
There are two target products. The first is "AVIEW IPN," which automatically detects pulmonary nodules from cardiac CT (CCTA) images, and the second is "AVIEW Lung Metrics," a quantitative analysis solution for lung diseases. Both products aim to obtain FDA 510(k) approval and to be commercialized in the U.S. market.
AVIEW IPN is a solution that automatically detects pulmonary nodules in lung regions included in cardiac CT scan images. It analyzes pulmonary findings—which are often deprioritized in cardiac-focused reading environments—without the need for additional examinations.
AVIEW Lung Metrics features the application of quantitative indicators (cLAA, cHAA) that are adjusted for variability based on scan conditions. This addresses the limitations of conventional quantitative analysis indicators, which were affected by scan protocols and noise, and enables consistent results across various equipment and imaging environments. According to the company, this technology could be utilized for global clinical trials in multinational and multicenter settings where imaging data reproducibility is required, as well as for image-based therapeutic response monitoring for treatments of chronic obstructive pulmonary disease (COPD) and idiopathic pulmonary fibrosis (IPF).
Currently, Coreline Soft has regulatory and clinical experience in 21 countries. The cumulative number of FDA 510(k) approvals stands at 12.
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- 20 Deaths Linked to US Vasculitis Drug in Japan Raise Safety Concerns
- "Target Price Set at 970,000 Won"... Top Investors Already Watching, Only an 'Uptrend' Remains [Weekend Money]
Meanwhile, Coreline Soft will participate in the American Thoracic Society (ATS 2026) conference to be held in Orlando, United States, from May 15 to 20, where it plans to showcase its AI-based workflow platform for lung disease analysis.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.