SK Bioscience-AZ, Vaccine Production Contract Until Year-End... "Extension Status Uncertain"
Task Force "AZ Vaccine to be Administered Only Until This Year"
SK Bioscience "Even if Extension Fails, Other Pharma Companies' Love Calls Continue"
SK Bioscience Accelerates GBP510 Clinical Trials Following Novavax Approval Application

SK Bioscience's 'L House Vaccine Center' in Andong-si, Gyeongbuk (Photo by SK Bioscience)
View original image[Asia Economy Reporter Lee Chun-hee] The current contract for the contract manufacturing organization (CMO) of AstraZeneca (AZ) COVID-19 vaccines, which SK Bioscience is producing, will expire at the end of this year. However, the decision on whether to extend the contract has not yet been finalized.
According to industry sources on the 18th, AZ announced on the 17th (local time) that "the CMO contract with SK Bioscience will end at the end of this year." SK Bioscience also confirmed that the CMO contract will expire at the end of this year.
SK Bioscience and AZ signed a COVID-19 vaccine CMO contract in July last year and have been producing vaccines at SK Bioscience L House in Andong, Gyeongbuk Province. The vaccine used for the first domestic inoculation on February 26 was also the AZ vaccine.
However, amid controversies over side effects such as thrombosis with thrombocytopenia syndrome (TTS), the proportion of vaccinations using the viral vector AZ vaccine has gradually decreased. The COVID-19 Vaccination Response Promotion Team announced yesterday that vaccinations using the AZ vaccine are planned to continue only until the end of this year.
Due to these circumstances, the possibility of extending the AZ vaccine CMO contract has entered an uncertain state. However, neither party completely denies the possibility of contract renewal next year. AZ stated, "We are continuing discussions about future plans and hope for a proactive review," and a SK Bioscience official said, "It is true that the current contract runs until the end of this year, but it has not yet been decided whether to extend or terminate the contract beyond next year."
Nevertheless, SK Bioscience believes that even if the contract with AZ ends, there will be no significant impact such as a decrease in sales. A SK Bioscience official emphasized, "Realistically, there are very few places capable of contract manufacturing vaccines, and we are receiving continuous love calls from many overseas pharmaceutical companies."
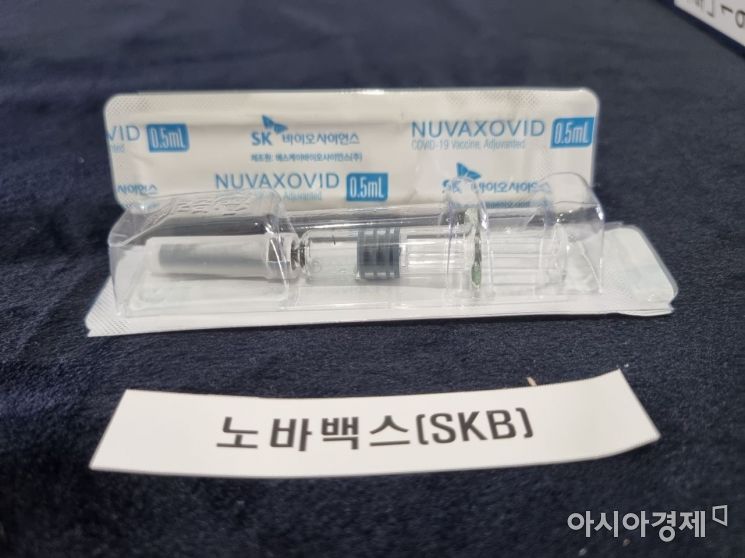
The Novavax COVID-19 vaccine, contract developed and manufactured (CDMO) by SK Bioscience, is exhibited at the Ministry of Food and Drug Safety on the 17th. / Photo by Lee Chunhee
View original imageMeanwhile, SK Bioscience applied for product approval for another contract development and manufacturing organization (CDMO) vaccine, the Novavax vaccine, on the 15th. It is a recombinant protein vaccine that injects the COVID-19 virus antigen protein into the body to generate antibodies.
SK Bioscience holds all domestic production and commercialization rights for the Novavax vaccine. After signing a CDMO contract with Novavax in August last year, the company also signed a technology transfer agreement in February to produce both the active pharmaceutical ingredient and finished drug product (DS).
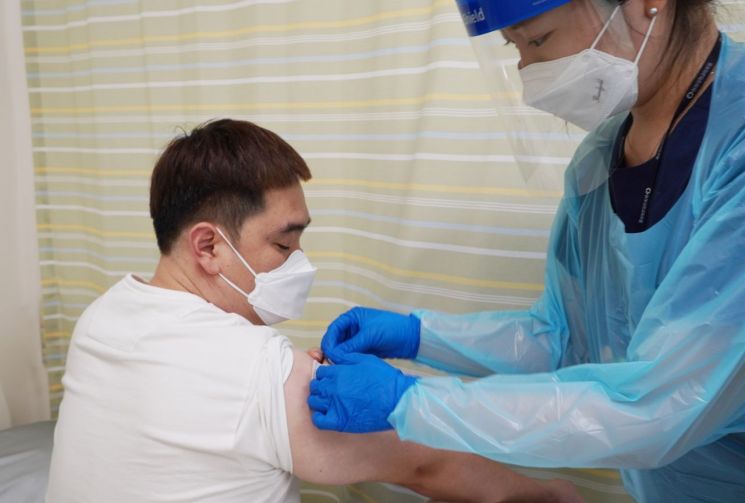
In August, subject administration for the Phase 3 clinical trial of SK Bioscience's COVID-19 vaccine candidate 'GBP510' was conducted at Dong-A University Hospital in Busan. (Photo by SK Bioscience)
View original imageSK Bioscience is currently conducting Phase 3 clinical trials for the COVID-19 synthetic antigen vaccine candidate 'GBP510,' which is being co-developed. More than 500 clinical participants have been administered the vaccine domestically, with many having completed the second dose. The company plans to request product approval from regulatory authorities around the first quarter of next year, aiming for product approval and launch in the first half of next year.
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
Yesterday, Kim Gang-rip, Commissioner of the Ministry of Food and Drug Safety, also expressed expectations that 'GBP510' will show good results in Phase 3 clinical trials, given the positive outcomes in Phases 1 and 2. Commissioner Kim stated, "There are clear limitations in predicting Phase 3 results based solely on Phases 1 and 2 results, but the data showed very satisfactory effects in terms of neutralizing antibody formation among clinical participants, and the neutralization potency of the formed antibodies was at an encouraging level." However, he added, "When the Phase 3 results are available, a final judgment will be made based on those figures."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







