Kim Kang-lip: "Vaccine approval proceeds independently of foreign countries"..."SK Bioscience vaccine satisfactory up to Phase 2" (Comprehensive)
"MFDS: Pharmaceutical Companies Must Apply for Approval to Proceed with Procedures"
"Novavax Can Be Approved Regardless of Foreign Authorization Status"
"GBP510 Shows Promising Results Up to Phase 2 Clinical Trials"
"Other Pharmaceutical Companies Also Discussing Clinical Trials with MFDS"
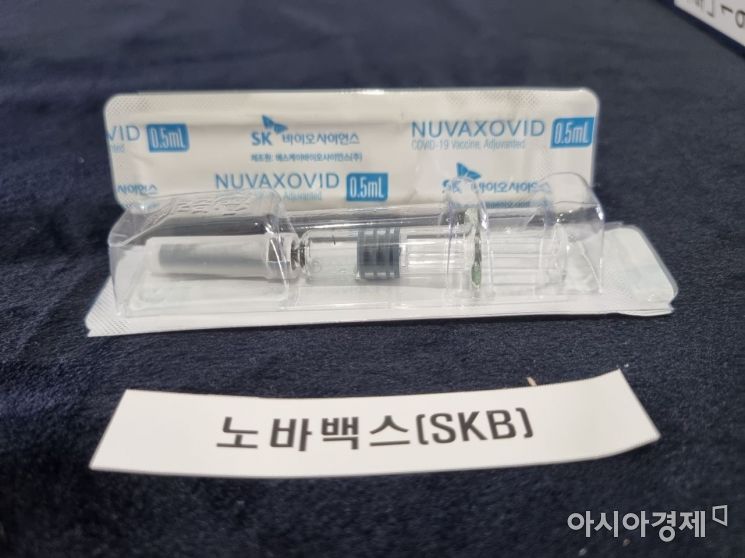
The Novavax COVID-19 vaccine, contract developed and manufactured (CDMO) by SK Bioscience, is displayed at the Ministry of Food and Drug Safety on the 17th. / Photo by Lee Chunhee
View original image[Asia Economy Reporter Lee Chun-hee] Kim Gang-rip, Commissioner of the Ministry of Food and Drug Safety (MFDS), strongly refuted criticisms that domestic vaccine approval procedures are conducted regardless of foreign approvals and that "follow-the-leader" approvals are taking place. Regarding the development of domestic vaccines, he said it is difficult to make premature judgments since only Phase 2 clinical trial results are available, but he evaluated that SK Bioscience is showing satisfactory results.
At a press briefing held at the MFDS in Osong, Chungbuk, on the afternoon of the 17th, Commissioner Kim clearly drew a line against criticisms that "domestic approval procedures only begin after overseas approval." He emphasized, "It is not true that domestic approval procedures are completed only if foreign approval is a prerequisite," and added, "(The MFDS) is the approval authority, and if companies do not proceed with the approval process, we cannot proceed on their behalf."
He acknowledged that he is aware of such criticisms and said, "(Vaccine developers) employ global strategies, so they strategically choose whether obtaining the first approval from the Korean MFDS will help their marketing, among other considerations." He stressed that this is entirely the vaccine developers' choice.
Accordingly, Novavax also expressed its intention to proceed with the approval review regardless of whether it has received approval from the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA). Commissioner Kim said, "If the given data is thoroughly reviewed and it is judged that there are no safety or efficacy issues and the vaccine meets the standards for approval, it can be approved regardless of foreign approvals. Although we are monitoring how approvals for the Novavax vaccine are progressing in other countries, foreign approvals do not directly affect our approval decision," he stated.
Currently, the Novavax vaccine has only received emergency use authorization in Indonesia. Novavax has applied for emergency use authorization from the World Health Organization (WHO), the European Union (EU), the United Kingdom, Canada, Australia, India, and the Philippines. The day before, SK Bioscience, which is conducting contract development and manufacturing organization (CDMO) work for the Novavax vaccine, applied for product approval of the Novavax vaccine.

SK Bioscience's 'L House Vaccine Center' in Andong-si, Gyeongbuk (Photo by SK Bioscience)
View original imageMeanwhile, regarding domestic vaccine development, SK Bioscience's 'GBP510' is currently undergoing Phase 3 clinical trials, and since it showed good results in Phases 1 and 2, it was evaluated as promising.
Commissioner Kim said, "It is certainly limited to predict Phase 3 results based solely on Phases 1 and 2 results," but added, "If we look only at Phases 1 and 2 results, applying the evaluation criteria that everyone vaccinated should form antibodies and that higher antibody levels indicate better vaccine functionality, the data showed very satisfactory effects broadly, and the horizontal criteria showed figures at a level worth expecting." However, he added, "When Phase 3 results come out, final judgments should be made based on those figures."
On the 5th, SK Bioscience disclosed the Phase 1 and 2 clinical trial results of GBP510, which it co-developed with the Antigen Design Research Institute (IPD) at the University of Washington School of Pharmacy in the United States. According to these results, GBP510 induced neutralizing antibodies that neutralize the COVID-19 virus in over 99% of the vaccinated group. Additionally, the level of neutralizing antibody induction was 3.6 to 13.3 times higher compared to the serum panel of COVID-19 convalescent patients. In terms of safety, no serious adverse reactions related to vaccination occurred.
Commissioner Kim said, "It is understood that about 500 participants in the domestic Phase 3 trial of GBP510 have completed their second dose, and vaccination clinical trial participants in Vietnam have also been vaccinated. The company plans to review conditional approval in the first quarter," he conveyed.
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
In August, subject administration for the Phase 3 clinical trial of SK Bioscience's COVID-19 vaccine candidate 'GBP510' was underway at Dong-A University Hospital in Busan. (Photo by SK Bioscience)
View original imageRegarding other domestically developed vaccines, he said, "One company is seriously consulting with the MFDS to enter Phase 3," and added, "We are working to enable Phase 3 entry as soon as possible, and we expect to enter Phase 3 soon." Currently, EuBiologics has submitted the Phase 3 trial plan for 'EuCorVac-19' to the MFDS, and Cellid has submitted the Phase 2b and 3 trial plans for 'AdCLD-CoV19-1.'
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.








