Examining Molecules One by One in Detail with Magnetic Resonance Imaging Device
-IBS Quantum Nano Science Research Group Achieves Single Molecule Electron Spin Resonance (ESR) Measurement
-Observation and Control of Spin Inside Single Molecule, Expanding Possibilities for Quantum Devices and Applications
[Asia Economy Reporter Kim Bong-su] Domestic researchers have succeeded in observing the state of a single molecule using the principle of magnetic resonance imaging (MRI) scanning.
The Institute for Basic Science (IBS) announced on the 12th that the Quantum Nano Science Research Group team succeeded in measuring the electron spin resonance (ESR) of molecules on a surface using a scanning tunneling microscope (STM). Electron spin resonance is a technology used to identify unknown molecules based on a principle similar to that of magnetic resonance imaging (MRI) commonly encountered in hospitals. This achievement is significant as it presents a new way to control the quantum states of molecules and secures fundamental technology for researching quantum phenomena based on molecules.
Recently, interest in utilizing individual spins in devices has increased due to the miniaturization of information processing devices. To create quantum devices from atoms or molecules, it is essential to perfectly control the interaction between two spins. For this, accurately understanding the spin of a single molecule is indispensable. In 2019, the research team developed a technology combining scanning tunneling microscopy and electron spin resonance to observe the magnetic field of a single atom, but there were limitations in understanding interactions within a single molecule or between single molecules. This was because the measurement sensitivity was low, causing multiple spins to be observed as a cluster.
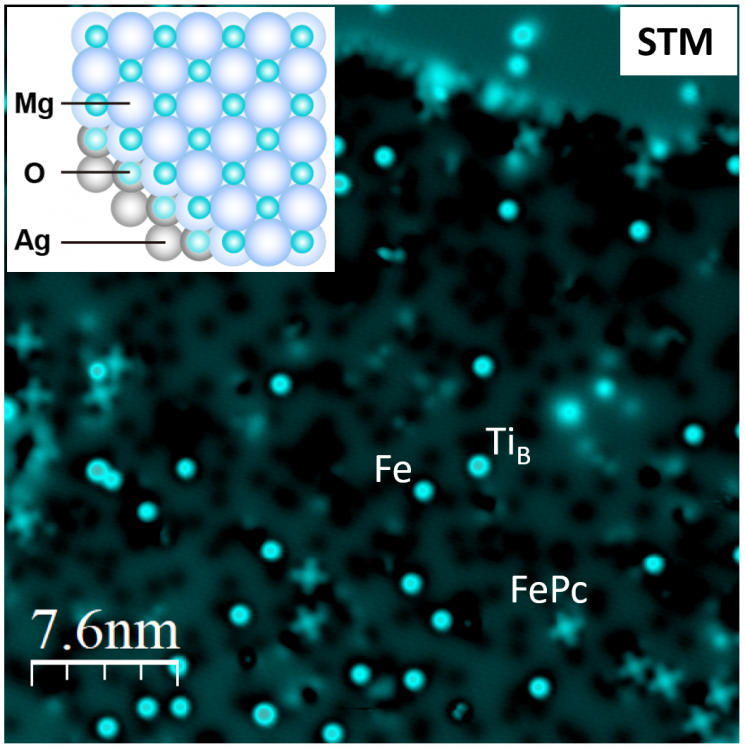
The research team focused on the characteristics of single iron (Fe) atoms, which are mainly used in scanning tunneling microscopy, and stably measured the electron spin resonance and magnetic interactions of single atoms and molecules. The experimental subject, iron phthalocyanine (FePc), is a ring-shaped organic compound with an iron atom at its center, widely used in organic solar cells, nanostructure synthesis, and chemical catalysts. The team deposited iron phthalocyanine (FePc) molecules, iron (Fe) atoms, and titanium (Ti) atoms on a magnesium oxide (MgO) insulating film on a silver (Ag) substrate and observed the subtle magnetic interactions of molecules and atoms using electron spin resonance phenomena.
This research is significant in that it expanded the measurement target of electron spin resonance from atoms to molecules. Molecules have much greater scalability compared to single atoms and have infinite potential for chemical applications. Previous studies measuring electron spin resonance of molecules had difficulty visualizing which molecule was being measured. However, this technology allows molecules to be directly seen and controlled, enabling applications in creating nanostructures as desired.
In particular, it is expected to greatly aid research in quantum sensing and quantum information science by enabling control of quantum-coherent states and quantum properties of spins. This measurement technology can be widely applied in magnetic material research, molecular structure research in medicine, and quantum sensing research because it can attach atoms or molecules with spins inside molecules whose chemical structures are unknown to identify their structures.
Research Fellow Jang Soo said, “When studying quantum states at the atomic scale, understanding the properties of single molecules is very important, and this research is a meaningful technology for measuring the magnetic interactions of molecules.” He added, “It will be an important stepping stone for developing molecule-based spin devices and quantum devices.”
Hot Picks Today
![Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416161763554_1778742977.png) Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- "Wearing a Leather Jacket in 30-Degree Heat, Jensen Huang Enjoys Street Food as Beijing's 'Mukbang Star': 'It's Delicious'"
- "Drink Three Cups of Coffee and Stay Up All Night Before the Test"... Manual of Insurance Planner Who Collected 1 Billion Won in Payouts
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
The research results were published on the 12th in the international journal Nature Chemistry (IF=24.427).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.