Discovery of 'Regulators' for Chronic Kidney Disease... Healing Cells
Identification of Key Cell Types and Regulators in Chronic Kidney Disease
Potential for Developing Drugs Specifically Targeting Certain Cell Types
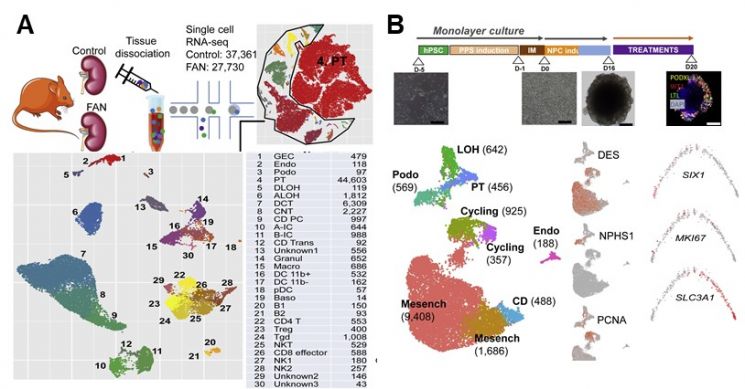
Single-cell analysis of kidneys from animal models induced with human chronic kidney disease (Figure A) and single-cell analysis of human kidney organoids (Figure B)
View original image[Asia Economy Reporter Junho Hwang] A study has been published that analyzed the kidney, which is composed of the most complex types of cells in our body, at the single-cell level and identified key regulators that play a crucial role in kidney development and the progression of chronic kidney disease. Through this, the research team suggested new therapeutic possibilities for chronic kidney disease. The research team led by Professor Jihwan Park of the Department of Life Sciences at Gwangju Institute of Science and Technology conducted an international joint study with the University of Pennsylvania in the United States and the Barcelona Institute of Science and Technology in Spain, and recently published the results in the international academic journal Cell Metabolism, the team announced on the 5th.
The research team identified important cell types involved in the progression of chronic kidney disease through the latest single-cell analysis technology and discovered the estrogen-related receptor (ESRRA), which plays a central role in the differentiation and metabolic regulation of these cells. Single-cell analysis technology is a technique that can analyze the expression of all genes expressed within tens of thousands of individual cells in a single experiment.
Through single-cell analysis of animal models induced with human chronic kidney disease and kidney organoids, the research team observed that many misdifferentiated proximal tubular epithelial cells exist in the kidneys during chronic kidney disease. They also found that there were problems in the lipid metabolism processes of these cells. Furthermore, ESRRA was identified as the key regulator controlling the differentiation and lipid metabolism of these proximal tubular epithelial cells, and it was confirmed that chronic kidney disease can be controlled by either inhibiting or overexpressing this gene.
Professor Jihwan Park said, "The greatest significance of this study lies in revealing the key cell types and regulators of chronic kidney disease by integrating the latest technologies such as single-cell analysis and kidney organoids," adding, "We confirmed the possibility of developing drugs that specifically target certain cell types through gene expression regulation in the future."
Hot Picks Today
!["Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]](https://cwcontent.asiae.co.kr/asiaresize/93/2025050713592847164_1746593968.jpg) "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
"Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Do We Need to Panic Buy Again?" War Drives 30% Price Surge... Even the Bedroom Feels the Impact
- "As Soon as We Hit Something, It Burst"... 13 Cars Damaged While Driving at Night
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Jay Y. Lee Bows His Head: "I Will Take All the Blame"... Apologizes for Samsung Labor-Management Conflict
Meanwhile, chronic kidney disease is a condition commonly accompanied by diabetes and hypertension. It is a disease whose number of patients is rapidly increasing worldwide, reaching approximately 800 million. Especially when it progresses to end-stage kidney disease, there is no treatment other than dialysis and kidney transplantation, causing great suffering to patients and their families, and the socioeconomic burden of the disease is also significant. Once chronic kidney disease begins to progress, there is currently no treatment to stop or reverse it, making the development of new therapeutics urgent.
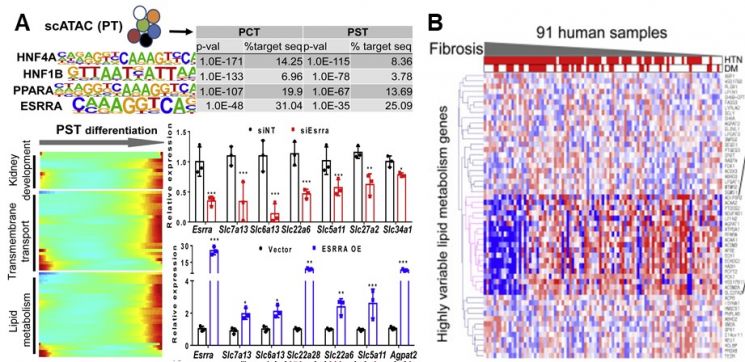
As a result of identifying the key gene ESRRA in chronic kidney disease through integrated analysis of single-cell gene expression and DNA structure (Figure A), and confirming the effect of ESRRA in a large human cohort dataset of 91 individuals (Figure B)
View original image© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







