Janssen·Pfizer Contracts This Month, Moderna Vaccine Next Month... "Domestic Vaccination in Q1" (Comprehensive)
Priority Vaccination Recipients to Begin Shots in February-March... Completion by Q4 Next Year
[Asia Economy Reporter Seo So-jeong] In response to the spread of the novel coronavirus infection (COVID-19), the government is accelerating COVID-19 vaccination efforts. To secure vaccines, contracts are being pursued this month with global pharmaceutical companies Janssen and Pfizer, and with Moderna targeting January next year, with plans to begin vaccinations as early as the first quarter.
On the 18th, the Central Disaster and Safety Countermeasures Headquarters (CDSCH) held an online briefing regarding the procurement of overseas-developed COVID-19 vaccines, announcing plans to sequentially introduce vaccines from February to March next year and start vaccinations promptly. Lim In-taek, Director of the Health Industry Policy Bureau at the Ministry of Health and Welfare, stated, "Considering the national crisis situation, we will designate and implement temporary vaccinations," adding, "We will prepare detailed execution plans for vaccination in December and thoroughly prepare to ensure rapid vaccination following the initial introduction (February to March)."
Director Lim explained, "We plan to complete vaccinations for recommended target groups by the fourth quarter of next year, before the influenza season," and added, "The target groups and detailed schedules will be flexibly adjusted considering supply volume, domestic COVID-19 situation, foreign vaccination status and side effects, and public demand."
Vaccination with AstraZeneca Vaccine to Start in First Quarter
Negotiations Ongoing with Novavax in Addition to Four Vaccines
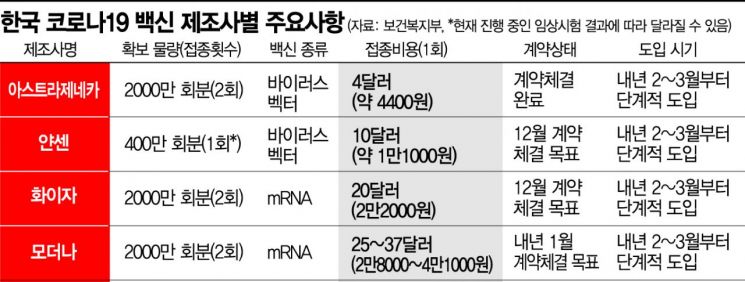
The government announced on the 8th that it has secured vaccines for up to 44 million people through global pharmaceutical companies and multinational alliances. Through the international vaccine development and supply project 'COVAX Facility,' 10 million doses will be secured, and through individual negotiations with multinational pharmaceutical companies such as AstraZeneca, Pfizer, Janssen, and Moderna, 34 million doses will be secured.
First, the purchase contract with AstraZeneca (20 million doses) has been completed. Contracts with Janssen (4 million doses) and Pfizer (20 million doses) are being pursued for December, and Moderna (20 million doses) for January. Director Lim said, "The contract with Janssen is expected to be completed as early as next week," and added, "The contract with Pfizer is currently in the final review stage, including legal review."
The vaccine expected to be administered the earliest is the AstraZeneca vaccine. It is the only vaccine for which the final contract has been completed so far, and the initial supply of AstraZeneca vaccines is produced under contract by SK Bioscience in Korea, making supply easier. Director Lim said, "AstraZeneca vaccine development is the most advanced, so negotiations started first procedurally and the contract was quickly finalized," adding, "The 'Made in Korea' vaccine from SK Bioscience is expected to be introduced around February to March next year, and preparations are underway to proceed with vaccinations according to the vaccination plan."
The government is also negotiating with Novavax in addition to AstraZeneca, Janssen, Pfizer, and Moderna. Director Lim said, "This morning, the government delegation held purchase negotiations with Novavax," adding, "There were discussions regarding quantities, but further negotiations are needed to determine the specific quantities that can be introduced."
Sequential Vaccination Starting with Priority Groups such as Healthcare Workers, Group Facility Workers, and the Elderly
The government plans to prepare detailed execution plans for vaccination within this month and start vaccinations as early as the first quarter. Priority groups for vaccination will include healthcare workers, residents and workers of group facilities, the elderly, adults with chronic diseases (ages 19?64, moderate or higher risk), workers and staff at child and youth education and care facilities, first responders to COVID-19, police officers, firefighters, and military personnel.
Vaccination for the general public is expected to be somewhat delayed. Yang Dong-gyo, Director of the Medical Safety and Prevention Bureau at the Korea Disease Control and Prevention Agency, explained, "The influenza season usually starts around November, and we are preparing with the goal of completing vaccinations for priority groups before then." Yang predicted, "Since priority groups cannot all be vaccinated at once, there may be sub-priorities within them. The priority groups will be vaccinated in stages, and the general public will be vaccinated after the priority groups' vaccinations are sequentially completed."
The government has been negotiating with Pfizer, AstraZeneca, Janssen, and Moderna since July to secure vaccines. Additionally, contract manufacturing (CMO) agreements were signed with AstraZeneca on July 21 and with Novavax on August 13. Considering the goal of securing vaccines for up to 44 million people and vaccination rates, the vaccine purchase cost is approximately 1.3 trillion KRW, which will be funded through the existing budget and next year's contingency funds.
The government is also discussing the introduction of vaccines secured through the COVAX Facility in the first quarter of next year. Director Lim said, "The supply timing for 10 million doses through COVAX is currently under discussion," adding, "At the initial announcement, three types of vaccines were proposed: AstraZeneca, Pfizer, and Sanofi-GlaxoSmithKline (GSK). However, Sanofi's vaccine development has been significantly delayed due to clinical issues, making it difficult to select Sanofi's vaccine."
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
Regarding domestic COVID-19 vaccine development, Director Lim said, "It is true that Korea's vaccine technology currently lags behind Western global pharmaceutical companies, but SK Bioscience is producing the AstraZeneca vaccine, and domestic development is being pursued jointly with Novavax," adding, "Hopefully, vaccines developed in Korea can be available by the end of next year or early the year after."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![[Image source=Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2020121814002673589_1608267626.jpg)