COVID-19 Vaccine Faces 'Triple Minefield' of Safety, Procurement, and Distribution Before February Inoculation Possible
Government "Plan for Rapid Vaccination in February-March Next Year"
Schedule Advanced but...
[Asia Economy reporters Seo So-jeong and Kim Heung-soon] The government plans to advance the COVID-19 vaccination schedule to enable vaccinations as early as the first quarter of next year, coinciding with the introduction of new coronavirus vaccines in February to March. This is due to the domestic COVID-19 cases remaining above 1,000 for three consecutive days without signs of slowing down, combined with seasonal factors indicating a prolonged outbreak. However, concerns have been raised that many challenges must be resolved before actual vaccinations can begin in February to March next year.
AstraZeneca's Ongoing Clinical Trials
Safety Issues Are the Biggest Obstacle
First and foremost, safety concerns are the biggest hurdle. The first COVID-19 vaccine to be administered in South Korea will be AstraZeneca's, but this vaccine is still undergoing Phase 3 clinical trials. Notably, serious side effects have been reported during the trials. In the UK, the clinical trial was temporarily halted in September due to an unexplained adverse event called transverse myelitis. After safety reviews, trials resumed in the UK in September and in the US in October. Similarly, Janssen, with which the government is finalizing contracts, temporarily paused its Phase 3 trials in October due to unexpected serious adverse events but has since resumed.
With AstraZeneca expected to receive emergency use authorization from the US Food and Drug Administration (FDA) in February next year rather than by the end of this year, there is also the burden of starting domestic vaccinations without FDA approval. On the 15th, Kwon Jun-wook, Deputy Director of the Central Disease Control Headquarters at the Korea Disease Control and Prevention Agency, stated at a regular briefing, "The FDA is a US agency, and South Korea conducts its own review through the Ministry of Food and Drug Safety," adding, "Regardless of FDA approval, the AstraZeneca vaccine will undergo review according to our country's procedures."
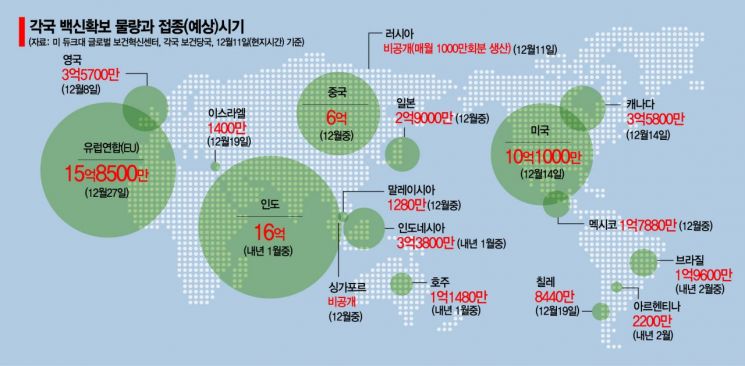
Given that AstraZeneca's clinical trials were halted twice due to serious side effects and that recent interim Phase 3 data showed varying efficacy depending on dosing methods, trust issues have surfaced, making safety a significant obstacle to vaccination. In response, Lim In-taek, Director of the Health Industry Policy Bureau at the Ministry of Health and Welfare, explained, "Rolling reviews of the AstraZeneca vaccine are ongoing in the UK and Europe, and safety has been confirmed through peer review in The Lancet," adding, "AstraZeneca has been pre-purchased in the largest quantities worldwide, with advanced countries such as the US (300 million doses), the UK (100 million doses), and Japan (120 million doses) also securing substantial amounts."
Different Distribution and Storage Requirements for Each Vaccine Pose Challenges
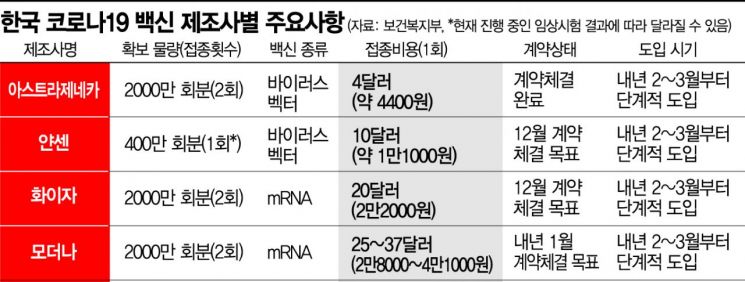
The four vaccines the government plans to introduce?Pfizer, Moderna, AstraZeneca, and Janssen?differ slightly in efficacy, storage methods, and shelf life, requiring thorough preparation. Based on immune response principles, Pfizer and Moderna are classified as messenger RNA (mRNA) vaccines. These work by injecting genetic material to produce antigens, which then stimulate antibody production. AstraZeneca and Janssen are viral vector vaccines, using harmless viral vectors to deliver genetic material that prompts the body to produce neutralizing antibodies capable of neutralizing the COVID-19 virus.
Particularly, Pfizer, which has already begun vaccinations in the US and UK, requires ultra-cold storage, making distribution and storage challenging. Yang Dong-gyo, Director of Medical Safety Prevention at the Korea Disease Control and Prevention Agency, stated, "The Pfizer vaccine must be stored at -60 to -80 degrees Celsius for six months and mixed with another liquid before administration," adding, "After thawing, it can be stored at 2 to 8 degrees Celsius for up to five days, so meticulous preparation is necessary." It should also be noted that two doses are required at a three-week interval.
The Moderna vaccine remains stable for up to 30 days at 2 to 8 degrees Celsius, typical of household or medical refrigerators, and can be stored for up to six months at -20 degrees Celsius. Vaccinations are administered twice at a four-week interval. AstraZeneca can be stored and handled at normal refrigeration temperatures of 2 to 8 degrees Celsius for at least six months, with two doses given four weeks apart. Janssen differs from the others by requiring only a single dose to elicit an immune response and can be stored at 2 to 8 degrees Celsius. Currently, vaccine efficacy rates have been disclosed as 95% for Pfizer, 94.1% for Moderna, 70% for AstraZeneca (up to 90% depending on dosing), and Janssen's efficacy is expected to be announced around January next year.
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
Director Yang added, "For ultra-cold frozen vaccines, manufacturers and importers are working on delivering them to vaccination sites while maintaining ultra-cold conditions, and vaccination centers are preparing to equip ultra-cold freezers for storage and management."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.