Government Decides on Special Import of COVID-19 Therapeutics... Timing of Securing Remains Uncertain (Update)
[Asia Economy Reporter Choi Dae-yeol] The government has decided to grant special import approval for Gilead's 'Remdesivir' from the United States as a treatment for the novel coronavirus infection (COVID-19). However, it is uncertain whether it can be immediately used at frontline medical institutions due to supply constraints.
On the 29th, the Central Disease Control Headquarters for COVID-19 announced that, reflecting the opinion of the Central Clinical Committee for Emerging Infectious Diseases, it will apply to the Ministry of Food and Drug Safety for special import approval of Remdesivir as an overseas pharmaceutical product. Special import approval is a system that allows the Ministry of Food and Drug Safety to bypass the approval or notification procedures for imported items when the head of the relevant ministry requests it in an emergency situation and the Ministry recognizes the urgent need for introduction.
Jung Eun-kyung, head of the Disease Control Headquarters, explained at the regular briefing that "Remdesivir has been evaluated to have safety and efficacy in treating COVID-19 pneumonia, and in the absence of alternative antiviral drugs, the medical community has agreed on the necessity of introducing Remdesivir."

Jung Eun-kyung, Director of the Central Disease Control Headquarters (Director of the Korea Disease Control and Prevention Agency), is holding a regular briefing on the afternoon of the 29th at the Korea Disease Control and Prevention Agency in Cheongju, Chungbuk, regarding the current status of domestic COVID-19 cases and the interim investigation results of confirmed patients.
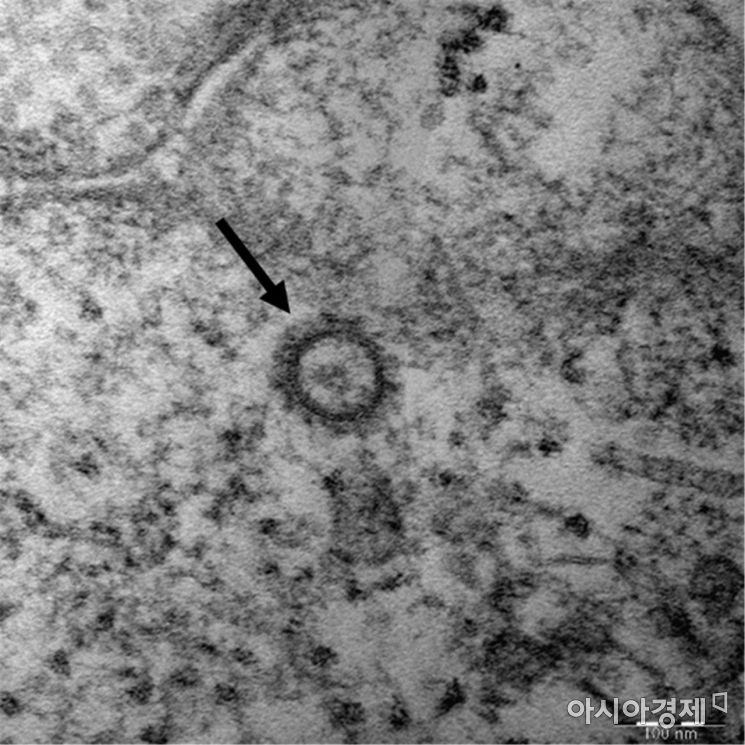
Remdesivir is a drug developed by the American pharmaceutical company Gilead Sciences originally for treating Ebola virus infection. In the case of COVID-19, recent global clinical trial results have demonstrated its efficacy in helping severe patients alleviate symptoms to mild levels and reduce hospitalization periods, leading to emergency use authorization in the United States. It is the first globally recognized COVID-19 treatment drug developed through drug repurposing by testing the effectiveness of existing drugs.
Head Jung stated, "It is targeted at patients with pneumonia who require oxygen therapy and have moderate to severe symptoms. The administration period is five days, which can be extended depending on the patient's condition, but monitoring for effectiveness and side effects is necessary. If the drug is introduced, usage guidelines will be created through monitoring indications, efficacy, and side effects."
However, even though the government has decided on special import approval, immediate supply is not guaranteed. Gilead is currently distributing Remdesivir mainly to countries that have approved and are using it, and supply is not abundant. Head Jung said, "We will proceed with two tasks: preparing usage procedures and guidelines, and securing the drug."
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.