MFDS: "4 COVID-19 Clinical Trial Plans Approved... 5 More Under Review"
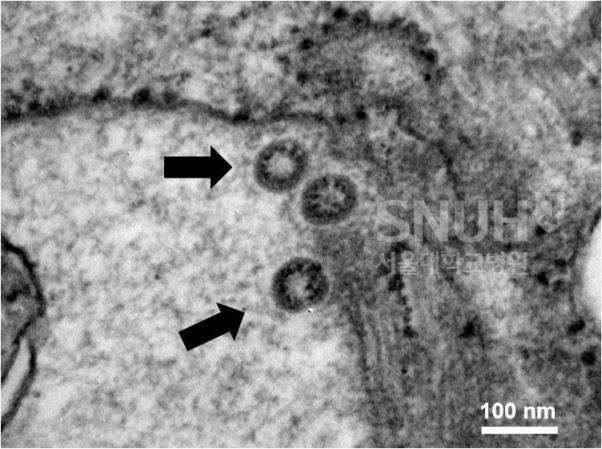
Seoul National University Hospital confirmed the COVID-19 virus using an electron microscope
[Asia Economy Reporter Choi Dae-yeol] While four clinical trials are underway for the application of treatments for the novel coronavirus infection (COVID-19), it has been revealed that, separately, five clinical trial plan applications have been submitted and are currently under review by regulatory authorities.
On the 24th, Yang Jin-young, Deputy Director of the Ministry of Food and Drug Safety (MFDS), stated at a briefing, "Regarding clinical trial plans related to COVID-19, four have been approved so far, and five other clinical trial applications have been submitted," adding, "Approval decisions will be made after thorough review."
According to the MFDS, two of the applications related to remdesivir, developed by the US-based pharmaceutical company Gilead Sciences as an Ebola treatment, were submitted by the company itself, and one by Seoul National University Hospital. Gilead is conducting a Phase 3 trial to evaluate safety and efficacy in severe and moderate patients, and Seoul National University Hospital is conducting a Phase 2 trial; both received approval in early this month. Additionally, Seoul Asan Medical Center received approval on the 20th to conduct an investigator-initiated clinical trial using Kaletra tablets, an HIV treatment, targeting mild patients.
Details regarding the specific institutions or drugs involved in the five applications currently under review have not been disclosed. As COVID-19 cases surge not only domestically but worldwide, active efforts are underway to verify whether existing drugs used for other diseases can be applied to COVID-19 patients alongside new drug development. The government is also actively supporting research and development domestically. However, the medical community anticipates that clinical trials must be conducted strictly following established procedures and regulations, and since outcomes are unpredictable, it will likely take considerable time before medical professionals can actually use these drugs.
Deputy Director Yang said, "To develop medicines for COVID-19 infection, we have formed a product development team within the Crisis Response Support Headquarters at the MFDS to actively consult with related companies and institutions regarding clinical trials and to listen to any difficulties," adding, "We will actively provide product development support consultations to expand patient treatment plans and facilitate the development of new therapeutics."
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







