Onconic Therapeutics Publishes Research on Next-Generation Anticancer Drug Candidate Nesuparip in International Journal
Onconic Therapeutics announced on May 12 that research findings related to its next-generation anticancer pipeline, nesuparip (JPI-547), have been published in the international oncology journal, the British Journal of Cancer (BJC).
The British Journal of Cancer is an SCI(E)-listed international journal in the field of oncology, published by Springer Nature, the world’s largest academic publisher. It is a Q1 (top 25%) journal with significant influence in cancer biology and anticancer drug development.
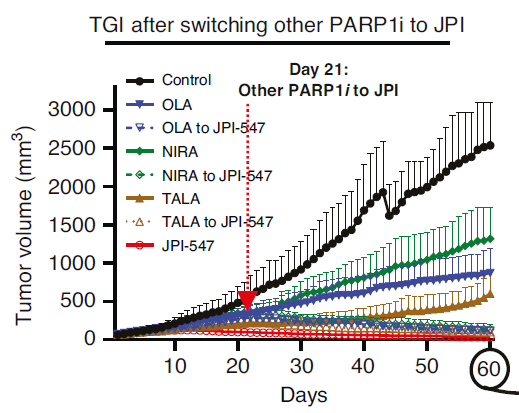
Graph comparing the antitumor inhibition of existing first-generation PARP inhibitors and Nesuparib (JPI-547) in a PARP-resistant model from the paper. Nesuparib monotherapy, shown in red, demonstrated stronger antitumor inhibition compared to the existing first-generation PARP inhibitors. Additionally, switching from existing PARP drugs to Nesuparib on day 21 resulted in increased antitumor inhibitory effects. Onconic Therapeutics
View original imageThis paper, led by Professor Moon Yonghwa’s research team from the Department of Oncology at CHA Bundang Medical Center (with Dr. Kang Minsil as the principal investigator), has drawn attention for analyzing the mechanisms behind resistance to existing PARP inhibitors such as olaparib, which are used in BRCA1/2-mutated cancers, and proposing the potential for new therapeutic strategies to overcome this resistance.
The research team established PARP inhibitor resistance models using BRCA-mutated breast and ovarian cancer cells and patient-derived tumor xenograft (PDTX) animal models, where PARP inhibitors are standard therapies. They reported that nesuparip demonstrated superior tumor suppression compared to existing PARP inhibitors.
According to the paper, nesuparip showed stronger tumor growth inhibition than the first-generation PARP inhibitor olaparib when administered alone in sensitive models responsive to olaparib. Notably, nesuparip alone also exhibited potent tumor growth suppression in resistant models where existing PARP inhibitors were ineffective. Furthermore, in both animal models, switching to nesuparip after existing PARP inhibitor administration led to a statistically significant increase in tumor regression, and in some cases, complete tumor regression was observed.
This study particularly focused on the RAD51 pathway, a key protein involved in DNA damage repair. The paper noted that in PARP inhibitor-resistant cancer cells, there was a tendency for increased RAD51 expression and restoration of homologous recombination (HR) DNA repair function.
Unlike existing PARP inhibitors, nesuparip functions as a dual inhibitor, simultaneously targeting PARP1/2 and tankyrase. By inhibiting the tankyrase-MERIT40-RAD51 axis, it reduced RAD51 expression and suppressed the recovery of HR DNA repair function. As a result, the anticancer efficacy of nesuparip was maintained even in models that had developed resistance to existing PARP inhibitors.
Additionally, the analysis of public data from ovarian and breast cancer patients revealed that groups with high RAD51 expression were more likely to experience cancer recurrence and lower survival rates. Based on these findings, the researchers suggested that the RAD51 pathway could be a potential biomarker associated with PARP inhibitor resistance. However, the company noted that this is a preclinical study, and additional clinical validation of efficacy in patients with high RAD51 expression is required.
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
The company stated, “This paper is meaningful as it suggests that nesuparip may regulate the RAD51 pathway, which is a key factor in PARP inhibitor resistance, going beyond simple antitumor efficacy. In fact, in our phase 1 clinical trial targeting multiple cancer types, more than 30% tumor reduction was observed in patients resistant to existing PARP inhibitors. We are therefore highly optimistic about nesuparip’s potential as a therapeutic strategy for patients with refractory cancers.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







