Molecular Mechanism Regulating Leukemia Drug Response Identified: "Clue to Overcoming Resistance"
The mechanism by which targeted anticancer agents induce the death of cancer cells has been identified. There is growing anticipation that utilizing this mechanism could enhance the effectiveness of treating patients who have developed resistance to targeted therapies.
On April 23, KAIST announced that a joint research team consisting of Professor Junghoon Lim from the Department of Biological Sciences at KAIST, Professor Dongwook Kim from the Hematologic Malignancy Center at Uijeongbu Eulji University Hospital, and Professor Hongtae Kim from Ulsan National Institute of Science and Technology (UNIST) has identified a novel molecular mechanism that regulates the response to chronic myeloid leukemia anticancer drugs.

(From left) Professor Dongwook Kim of the Blood Cancer Center at Uijeongbu Eulji University Hospital, Professor Hongtae Kim of UNIST, Professor Junghoon Lim of KAIST, and Dr. Joomin Park of KAIST. KAIST
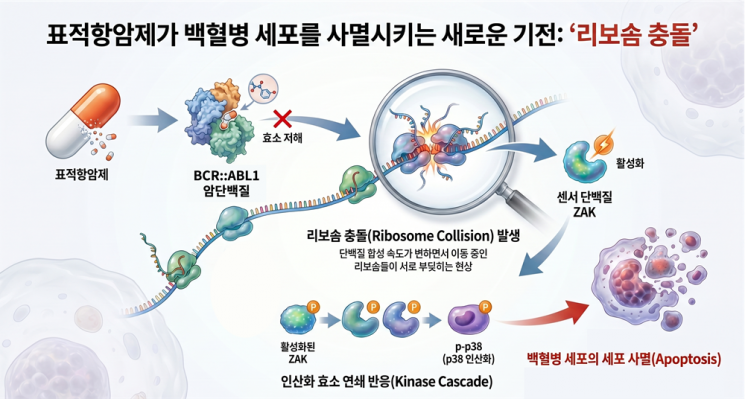
View original imageChronic myeloid leukemia is caused by a genetic abnormality in hematopoietic stem cells, leading to the production of an abnormal protein known as 'BCR::ABL1.' This protein is known to send growth signals to cells, which is a major cause of the continuous proliferation of cancer cells.
Targeted anticancer agents are used as the standard treatment method to inhibit this process. However, some patients exhibit drug resistance or low treatment response, resulting in limited effectiveness of targeted therapies.
In this context, the joint research team focused on the impact of anticancer agents on the intracellular protein production process. They discovered that when anticancer agents are administered, the flow of ribosomes—which produce proteins—becomes disrupted, causing ribosomes to collide, a phenomenon referred to as 'ribosome collision.' This process induces significant stress within the cell, ultimately leading to the self-destruction of cancer cells.
The key sensor that detects ribosome collision was identified as the 'ZAK' protein. Notably, the joint research team found that, under normal conditions, the ZAK protein binds with the AKT signaling pathway (a pathway that regulates cell survival, growth, proliferation, metabolism, and migration), thereby acting as a facilitator for cancer cell growth. However, once targeted anticancer therapy begins, ZAK switches roles to monitor ribosome collision and acts as a sentinel that triggers cancer cell death.
This is the first case in the world to prove that the ZAK protein plays completely opposite roles during the progression and treatment of cancer.
The joint research team also validated this mechanism by analyzing cancer cells derived from leukemia patients. The results showed that using drugs that increase ribosome collision enhances the anticancer effect, whereas decreased ZAK function leads to reduced responsiveness to anticancer agents.
Based on these findings, the team concluded that decreased ZAK function or insufficient ribosome stress response can induce resistance to targeted anticancer therapy. In other words, this suggests that it is possible to predict an individual patient's treatment response based on their ZAK activity level and to design personalized combination treatment strategies.
This study is significant in that it highlights the importance of the ribosome stress signaling pathway in the treatment of chronic myeloid leukemia. The joint research team expects that, in the future, these findings could lead to increased efficacy of targeted therapies and the development of new combination treatment strategies.
In particular, this may offer new therapeutic possibilities for patients who struggle with treatment due to drug resistance.
Professor Lim stated, "This study demonstrates how crucial it is for cells to detect abnormal protein synthesis and convert this into a death signal in the context of treatment."
Hot Picks Today
 Chilling Timing "Did They Know Again?"... $640 ...
Chilling Timing "Did They Know Again?"... $640 ...
- Thailand and India Surpass 50%, but Korea Only at 37%... Falling Behind in the A...
- Borrowed 6.8 Billion Won in Company Funds to Buy 11.75 Billion Won Home... Numer...
- "Woke Up to a Strange Smell"...Both Feet Placed on KTX Front Armrest
- "If You Have This at Home, Remove It Immediately"... 'This Item' Is as Harmful a...
Meanwhile, Dr. Jumin Park of KAIST participated as the first author of this study. The research findings were published online in the hematology journal 'Leukemia' on March 30.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.