"No Need for Platinum"…Yonsei University Develops 'Dual-Atom Catalyst' to Boost Zinc-Air Battery Performance [Reading Science]
Implementation of Dual Iron Active Sites via Polymer Interface Engineering
Enhancing Both Lifespan and Output
A non-precious metal catalyst with performance surpassing that of commercial platinum catalysts has been developed, raising the possibility of overcoming a key bottleneck in the commercialization of zinc-air batteries—a next-generation energy storage device. This technology is highly regarded for its industrial impact, as it can enhance performance while reducing reliance on expensive precious metal catalysts.
On March 19, a joint research team led by Professor Junghoon Kim from the Department of Chemical and Biomolecular Engineering and Professor Sungmin Park from the Department of Materials Science and Engineering at Yonsei University announced the development of a highly active and durable oxygen reduction catalyst by utilizing a dual iron (Fe) active site structure based on polymer interface engineering. The research results were published in the top-tier journal in the field of catalysis, Applied Catalysis B: Environment and Energy.
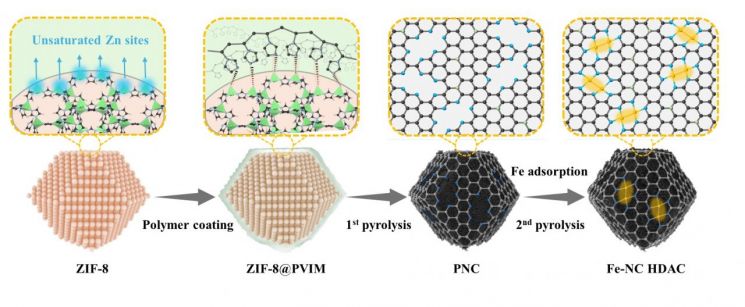
Design Strategy for High-Density Dual Iron Active Site Catalyst through Metal-Organic Framework-Based Interface Control. Photo by Research Team
View original image"Performance Exceeds Platinum"…Breakthrough for Non-Precious Metal Catalysts
Zinc-air batteries have been noted as a next-generation battery due to their high energy density and cost-effectiveness. However, they have faced limitations in performance because of the slow oxygen reduction reaction at the air electrode. In particular, conventional single-atom iron catalysts have struggled to achieve both activity and stability, as metal atoms tend to aggregate or the active structures are limited.
To address this challenge, the research team introduced a polymer interface control strategy. By uniformly coating a nitrogen-rich polymer onto the surface of a metal-organic framework (MOF) and then carbonizing it, they successfully formed adjacent dual iron active sites in a stable manner. This process not only suppressed metal aggregation but also achieved both high iron content and atomic-level dispersion simultaneously.
As a result, the developed catalyst recorded a half-wave potential of 0.91V, demonstrating superior oxygen reduction performance compared to commercial platinum (Pt/C) catalysts (0.86V). When applied to actual zinc-air batteries, it also achieved an open-circuit voltage of 1.46V, a maximum power density of 182mW/cm², and demonstrated stable charge and discharge performance for over 300 hours, thereby proving both high activity and durability.
"Cooperative Action of Dual Active Sites"…Proven Performance and Scalability
The research team also elucidated the mechanism behind the enhanced performance through density functional theory (DFT) calculations. They found that two adjacent iron active sites more efficiently activate oxygen molecules, and in particular, when the structure is pre-adsorbed with hydroxide ions, the reaction proceeds at a low overpotential (0.24eV).
This provides a more favorable reaction pathway compared to conventional single active site (FeN4) catalysts, demonstrating that the cooperative action between dual active sites is a key factor in boosting oxygen reduction reaction performance.

Research team photo. From left to right: Donggyun Kim, integrated graduate student in Chemical and Biomolecular Engineering; Sunghyun Jeong, integrated graduate student in Chemical and Biomolecular Engineering; Junghoon Kim, professor in Chemical and Biomolecular Engineering; Myungjun Song, student in Materials Science and Engineering; Sungmin Park, professor in Materials Science and Engineering; Godeung Park, integrated graduate student at Korea Electrotechnology Research Institute. Provided by Yonsei University
View original imageFurthermore, the team confirmed that the process maintains the same performance even at gram-scale synthesis, emphasizing that this is a meaningful achievement in terms of mass production potential.
Professor Junghoon Kim of the Department of Chemical and Biomolecular Engineering at Yonsei University stated, "The key is that we proposed an interface design strategy capable of stably realizing the dual active site structure," adding, "This approach can be expanded not only to next-generation metal-air batteries but also to a variety of energy conversion and storage systems."
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- International Oil Prices Plunge Over 7% on Hopes for US-Iran Deal...Largest Drop...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
This study was jointly conducted by first authors Donggyun Kim (PhD candidate), Sunghyun Jung, Godeung Park, and Myungjun Song, with theoretical calculations carried out by the Korea Electrotechnology Research Institute (KETI). The research was supported by the National Research Foundation of Korea’s Nano and Material Technology Development Program, the Industrial Innovation Talent Growth Support Program of the Korea Institute for Advancement of Technology, and the NRF LAMP program.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







