Producing Two Chemical Materials from a Single Glucose Molecule... Low-Carbon Catalyst Operates Without Hydrogen [Reading Science]
Successful Simultaneous Production of Gluconic Acid and Sorbitol
Room-Temperature Process Expected to Reduce Energy Use and Carbon Emissions
A domestic research team has developed a low-carbon catalyst technology capable of simultaneously producing two high-value chemical materials from a single glucose molecule. This reaction can proceed at room temperature without the need for external hydrogen supply or high-pressure equipment, presenting a new possibility for eco-friendly bio-chemical processes.
On March 15, the Korea Research Institute of Chemical Technology (KRICT) announced that the research team led by Principal Researcher Youngkyu Hwang, Senior Researcher Kyungyul Oh, and Senior Researcher Jihoon Kim has developed a cyclic low-carbon catalyst technology that enables the simultaneous production of gluconic acid—a raw material for detergents and pharmaceuticals—and sorbitol—a sweetener and cosmetic ingredient—from glucose.

The special catalyst developed by the research team at the Korea Research Institute of Chemistry (right), is made by loading a solution mixing platinum and tin in a ratio of 3 to 1 onto a support at 3% by weight of the support. Provided by KRICT
View original imageThe research findings were published this January in the international journal in the field of catalysis, "Applied Catalysis B: Environment and Energy."
Simultaneous Production of Two Materials by Utilizing Internal Hydrogen in Glucose
Gluconic acid and sorbitol are representative bio-based chemical materials with an annual global production of several million tons. Gluconic acid is used in detergents, pharmaceuticals, and metal cleaners, while sorbitol is utilized as a sweetener and cosmetic ingredient.
However, in existing processes, each product had to be manufactured separately from glucose as the raw material. In particular, manufacturing sorbitol required high energy consumption and costs, as well as significant carbon emissions, because it necessitated high temperatures above 100°C and high-pressure hydrogen above 10 atmospheres.
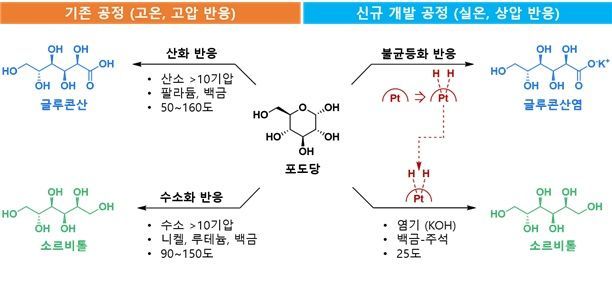
Comparison of New Eco-friendly Glucose Conversion Process Technology for Producing Gluconic Acid and Sorbitol Using Disproportionation Reaction with Existing Technologies. Previously, glucose had to be converted by either oxidation or hydrogenation reactions, requiring high temperatures above 100°C and high-pressure gases over 10 atmospheres, resulting in high process costs and environmental burdens. The research team developed a glucose disproportionation reaction that operates at room temperature and atmospheric pressure without external hydrogen or oxygen supply, internally recycling the hydrogen generated during the reaction to simultaneously produce gluconic acid and sorbitol. This technology can be expanded as an eco-friendly process applicable to various sugars, including galactose, mannose, and xylose. Provided by the research team
View original imageTo address these issues, the research team applied an "Internal Hydrogen Transfer" reaction that utilizes hydrogen within the glucose molecule. During the conversion of glucose to gluconic acid, the generated hydrogen is not released externally but is immediately transferred to another glucose molecule, converting it into sorbitol. As a result, both products can be produced simultaneously in a single reaction system without supplying external hydrogen or oxygen gas.
In experiments, when 100 glucose molecules were introduced, a selectivity of 50 to 50 was observed, resulting in the production of 50 gluconic acid molecules and 50 sorbitol molecules.
Enhanced Hydrogen Transfer Efficiency with Platinum-Tin Catalyst
The core of this process is a bimetallic catalyst combining platinum (Pt) and tin (Sn). The research team combined the two metals in a 3-to-1 ratio to precisely control the reaction rate.

Research team at Korea Research Institute of Chemical Technology. Top row from left: Senior Researcher Jihoon Kim, Postdoctoral Researcher Ali Awad, Senior Researcher Kyungyul Oh, UST Student Researcher Kirola Kolmizaeva, Principal Researcher Youngkyu Hwang. Provided by KRICT
View original imageIf only platinum is used, the reaction proceeds excessively fast, causing hydrogen loss as gas. However, when tin is used together, the reaction rate can be controlled, greatly improving hydrogen transfer efficiency. This enabled the implementation of a cyclic reaction system where the generated hydrogen is utilized in subsequent reactions.
Moreover, unlike previous studies conducted under low glucose concentrations of 1-5%, this research confirmed stable reactions even in a high-concentration glucose environment of 36%.
Industrial-Scale Productivity Achieved... Expectation for Eco-friendly Chemical Processes
The technology has also achieved productivity levels suitable for industrial applications. The research team recorded a daily product yield of about 1.5 kg or more per 1-liter reactor, confirming competitiveness compared to the existing high-temperature, high-pressure process.

Cover image of 'Applied Catalysis B' introducing research results. Provided by Korea Institute of Chemistry.
View original imageAfter the reaction, gluconic acid and sorbitol were separated at high purity using bipolar membrane electrodialysis, an electricity-based separation technology. The separation process cost was found to be economically favorable at around 150 won per kilogram of product.
This technology can be applied not only to glucose but also to the production of various bio-based chemical materials such as xylitol, using sugars derived from wood like xylose.
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- Woman in Her 50s Found Dead 28 Days After Going Missing on Bukhansan Mountain
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
Principal Researcher Youngkyu Hwang of KRICT stated, "This study presents a cyclic chemical process model that recycles internal hydrogen in glucose," adding, "By using plant resources instead of petroleum and significantly reducing carbon emissions, it will contribute to the transition to an eco-friendly chemical industry."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







