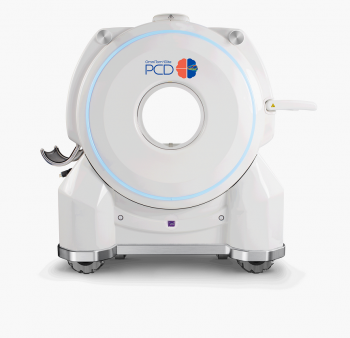
Samsung Neurologica Obtains FDA Additional Approval for 'OmniTom Elite PCD'
Strengthening Global Competitiveness of Mobile CT Products
Selected as 'Innovation Technology of the Year' in the USA
Samsung Electronics' U.S. medical device subsidiary NeuroLogica has been recognized for the technology and innovation of its mobile CT product equipped with a photon counting detector (PCD), the OmniTom Elite PCD.
On the 22nd, NeuroLogica announced that it had obtained approval from the U.S. Food and Drug Administration (FDA) on the 10th (local time) for new feature additions to the OmniTom Elite PCD. The approval was granted after adding ultra-high resolution mode, PCD-specialized applications, expanded scanning range, and continuous helical scan functions to the OmniTom Elite PCD.
The PCD applied to the OmniTom Elite is a next-generation CT scanning technology utilizing semiconductors. Compared to the conventional method using scintillators, it provides CT images with higher resolution and lower noise. Through the ultra-high resolution mode, it is now possible to adjust images so that residual contrast agents are not visible in CT scans taken after administering contrast agents.
NeuroLogica expects that the addition of FDA-approved new features to the OmniTom Elite PCD will strengthen its global competitiveness in the mobile CT field. The OmniTom Elite PCD was selected as the 'Innovation Technology of the Year' product by Premier. Premier is a hospital management and purchasing agency company with over 4,350 hospitals as members in the United States. Every year, it selects innovative companies and technologies based on technological innovation and contributions to improving medical accessibility within local communities.
Yoo Kyu-tae, head of Samsung Electronics' Medical Device Business Division and CEO of Samsung Medison, said, "We will continue to lead the development of new technologies in the medical imaging field and strive to enhance convenience for medical professionals and diagnostic accuracy."
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
Meanwhile, the new features of the OmniTom Elite PCD approved by the FDA are also awaiting CE MDR (European Union Medical Device Regulation) certification and approval from the Ministry of Food and Drug Safety. In Korea, approval for functions based on the existing FDA-approved features was obtained from the Ministry of Food and Drug Safety last year. Samsung Electronics' subsidiary Samsung Medison handles domestic sales and services of NeuroLogica's mobile CT products, including the OmniTom Elite PCD, as well as Samsung Electronics' digital X-ray devices.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.