Annual 50 Million Tons of Discarded Mokchoaek... Producing High-Value Chemical Compounds
KAIST Research Team
[Asia Economy Reporter Kim Bong-su] Domestic researchers have succeeded in producing high value-added compounds using wood tar produced when burning wood. Although more than 50 million tons are produced annually, most of the wood tar is discarded, making its recycling industrially noteworthy.
The Korea Advanced Institute of Science and Technology (KAIST) announced on the 28th that Professor Park Chan-beom's research team in the Department of Materials Science and Engineering identified the photocatalytic properties of lignin, a major component of plants, and successfully achieved artificial photosynthesis that produces high value-added compounds using sunlight by combining lignin-based photocatalytic reactions with redox enzyme reactions.
Lignin is one of the main components of "wood tar" produced when burning wood such as oak. It is a major substance forming the plant's woody tissue and is the second most abundant component after cellulose. It mainly serves as a structural framework supporting and protecting plants.
Lignin plays roles in cell wall formation, water transport, seed protection, and stress adaptation. In the wood industry producing biofuels, pulp, and paper, lignin is discharged as a byproduct in large quantities, reaching 50 million tons annually. However, due to its highly complex molecular structure, lignin is difficult to utilize and over 95% is incinerated or discarded.
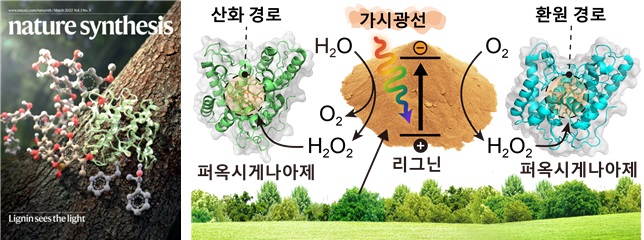
The research team hypothesized that lignin could function as a photocatalyst, inspired by the fact that natural lignin contains functional groups typical of common photocatalysts. They demonstrated that various lignin polymer models generate hydrogen peroxide under visible light. Spectroscopic and (photo)electrochemical analyses confirmed that lignin can thermodynamically induce photoredox reactions.
Typical photocatalysts require a sacrificial electron donor (such as alcohol, formic acid, or glucose) to reduce oxygen and generate hydrogen peroxide. Due to these requirements, existing photocatalytic reactions producing hydrogen peroxide have low atom economy and accumulate undesirable byproducts. However, lignin can synthesize hydrogen peroxide using oxygen and water without a sacrificial electron donor, showing high atom economy (94.4%) and avoiding byproduct accumulation.
Furthermore, the research team applied lignin's visible light-absorbing photocatalytic reaction to the biocatalyst peroxygenase activity. Peroxygenase is an enzyme that can induce highly selective oxyfunctionalization reactions important in organic synthesis. Although peroxygenase requires hydrogen peroxide, it is deactivated by high concentrations of hydrogen peroxide. To overcome this, the team designed lignin to photochemically generate hydrogen peroxide at an appropriate rate, enabling peroxygenase to continuously perform oxyfunctionalization reactions.
Professor Park Chan-beom stated, "This research is significant in presenting an eco-friendly method to utilize lignin for producing high value-added compounds," adding, "We plan to further elucidate the photocatalytic mechanism of lignin to enhance its catalytic performance, combine it with various enzymes, and produce fine chemical products to increase industrial impact."
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- "Target Price Set at 970,000 Won"... Top Investors Already Watching, Only an 'Uptrend' Remains [Weekend Money]
The research results were published as the cover paper in the March issue of the international journal Nature Synthesis.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.