Humasis, Overseas Export of COVID-19 Diagnostic Kits... Performance Also 'Soars'
The number of new COVID-19 cases is breaking record highs day after day. To respond to the spread of the Omicron variant, the government has shifted the COVID-19 diagnostic testing system to focus on self-diagnostic tests. The previously used polymerase chain reaction (PCR) tests are now prioritized for high-risk groups. There is growing anticipation that diagnostic companies such as SD Biosensor and Humasis, which have launched COVID-19 self-test kits, will benefit. Asia Economy examines the business structure, production capacity, and financial status of SD Biosensor and Humasis to gauge their growth potential.
[Asia Economy Reporter Park Hyungsoo] As new COVID-19 cases in South Korea continue to hit record highs, Humasis achieved its best-ever performance last year. By swiftly developing COVID-19 diagnostic kits and exporting them worldwide, its sales grew rapidly. With the government deciding to utilize self-test kits, Humasis's performance outlook remains bright this year as well.
According to the Financial Supervisory Service's electronic disclosure system on the 7th, Humasis recently signed a contract with the Public Procurement Service to supply COVID-19 self-test kits worth 24.2 billion KRW. This amount corresponds to 52.9% of its 2020 sales. The contract period extends until January 27 of next year.
As the Omicron variant spreads rapidly, the government, considering the limitations of PCR testing capacity, has prioritized rapid antigen testing. Individuals not classified as high-risk who visit screening clinics receive rapid antigen tests using self-test kits under supervision. Currently, test kits from three companies?SD Biosensor, Humasis, and Rapigen?have received official approval.
The antigen diagnostic kits and self-test kits developed by Humasis are known to detect the Omicron variant as well. At the end of last year, in collaboration with a local national university in South Africa, an evaluation was conducted on 30 clinical specimens infected with the Omicron variant. The evaluation showed a sensitivity of 93.3%. A Humasis representative stated, "Humasis products have been confirmed to detect the Omicron variant without any difference from the existing COVID-19 virus detection methods."
Founded in 2000, Humasis is a developer of in vitro diagnostic devices. It has focused its research capabilities on point-of-care diagnostics within in vitro diagnostic testing. Thanks to this, it has succeeded in commercializing diagnostic reagents of various formulations and quantitative analyzers. It has secured core technologies for equipment, reagents, and raw materials for point-of-care diagnostics. The company has strengthened its competitiveness by building a rapid response system for infectious diseases, ensuring user convenience and test accuracy. It has filed overseas patents for test products related to cardiovascular diseases, infectious diseases, and obstetrics and gynecology, earning recognition for its technological capabilities.
Cha Jeonghak, the largest shareholder and CEO, earned a master's degree in genetic engineering from Korea University in 1990. He worked as a researcher at Yuhan Corporation until 2000, gaining experience in pharmaceutical research and development. After joining Humasis, he served as head of the central research institute and was appointed CEO in 2009.
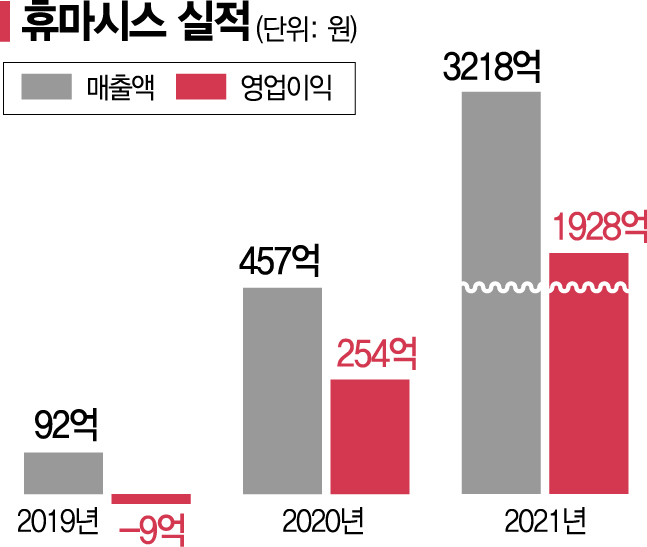
Although the company experienced continuous losses until 2019, sales surged significantly after developing COVID-19 diagnostic kits in 2020. Sales increased from 9.2 billion KRW in 2019 to 45.7 billion KRW in 2020 and 321.8 billion KRW in 2021. Operating profit was 25.4 billion KRW in 2020 and 192.8 billion KRW in 2021. In the fourth quarter of last year, sales reached 173.9 billion KRW with an operating profit of 98.8 billion KRW.
The sharp improvement in performance is likely to continue for the time being. On the 22nd of last month, Humasis signed a contract worth 136.6 billion KRW with Celltrion to supply COVID-19 antigen diagnostic home kits. This amount is three times its 2020 sales. The contract period runs until April 30 of this year.
Celltrion USA, Celltrion's American subsidiary, will supply approximately 400 billion KRW worth of COVID-19 rapid antigen test and diagnostic kits to the U.S. by April. The products supplied include the 'DiaTrust COVID-19 Professional Antigen Rapid Diagnostic Kit' jointly developed by Celltrion and Humasis, and the general home-use 'DiaTrust COVID-19 Antigen Home Test.' Celltrion USA is also negotiating additional quantities with major clients for the second quarter, which could increase Humasis's supply volume.
As performance improves, the financial structure has also strengthened. As of the end of the third quarter last year, cash equivalents stood at 74.6 billion KRW, exceeding total liabilities of 38.8 billion KRW. With large profits recorded in the fourth quarter, investment capacity has further increased. Humasis's production facility utilization rate was 86% in the third quarter. Considering quarterly sales and supply contracts, expansion investments are likely to continue. The largest shareholder's stake is only 7.58%.
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.