UNIST·KAIST Joint Research Team Develops 'Catalyst-Controlled Synthesis Method' by Splitting Expensive Precious Metals
'Reactivity Control of Single-Atom Catalysts ... Development and Application of Various Catalysts with Reduced Precious Metal Content'
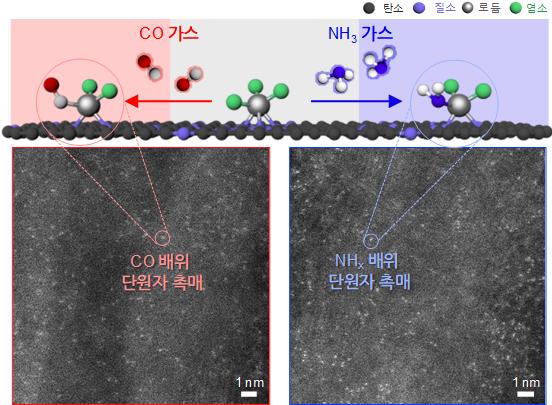
Schematic diagram of coordination structure control and microscopic image of coordination structure control catalyst.
View original image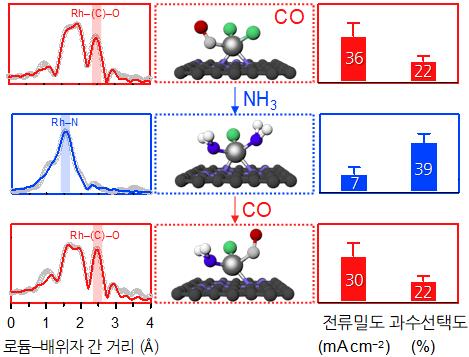
A research figure showing changes in catalyst structure and reactivity according to ligand modulation of rhodium single-atom catalysts.
View original image[Asia Economy Yeongnam Reporting Headquarters Reporter Kim Yong-woo] A domestic research team has jointly developed a single-atom catalyst synthesis technology that allows precious metals to be finely divided and used.
This is a ‘hidden gem’ technology that can reduce costs by lowering the content of precious metals and using them sparingly.
Ulsan National Institute of Science and Technology (UNIST) announced on the 30th that it has newly developed a single-atom catalyst synthesis technology that can finely divide precious metals in collaboration with KAIST and the Pohang Accelerator Laboratory joint research team.
This is a synthesis method that allows control of the reactivity of single-atom catalysts. This method can reduce the amount of expensive precious metals used in catalysts and is expected to be applicable to the development of various catalysts.
The research led by Professor Joo Sang-hoon’s team at UNIST’s Department of Chemistry involved Professor Kim Hyung-joon’s team from KAIST’s Department of Chemistry and Dr. Lee Guk-seung’s team from the Pohang Accelerator Laboratory.
Single-atom catalysts are catalysts in which metal atoms are dispersed one by one on the surface of a support. Since each metal atom can be used as a reaction site, it is more efficient even when using the same weight of precious metals.
However, controlling the reactivity such as catalytic activity and selectivity of single-atom catalysts is challenging because it is difficult to freely adjust the types of ligands, molecules or ions surrounding the central metal.
The reactivity of single-atom catalysts is highly sensitive to the coordination structure formed by the bonding of the central metal and ligands.

(From left) UNIST Researcher Jinjong Kim, Researcher Junsung Lim, Dr. Jaehyung Kim.
View original imageProfessor Joo’s research team developed a single-atom catalyst synthesis method that easily changes the type of ligand.
This method exchanges the ligands remaining on the metal precursor (raw material) during the catalyst synthesis process with other types of ligands.
When the residual ligand (CO) bonded to rhodium (Rh), a platinum-group precious metal, was exchanged with another ligand (NHx), the catalytic activity (reaction rate) for the oxygen reduction reaction differed by up to 30 times, and the reaction selectivity showed a threefold difference.
Single-atom catalysts are synthesized by fixing metal precursors surrounded by ligands onto a support and then removing the precursor ligands.
Previously, many attempts were made to change the coordination structure by adjusting the bonding between the metal and the support.
Also, the newly developed synthesis method allows ligands to be repeatedly exchanged (reversible reaction), enabling re-adjustment of the controlled coordination structure.
First author Dr. Kim Jae-hyung explained, “Unlike the conventional method that requires multiple trials and errors to achieve the desired reactivity, this study demonstrated that the coordination structure can be controlled as desired by changing the residual ligands of the metal precursor.” He added, “It can also be applied to catalyst development using various metal elements and support materials.”
Dr. Lee Guk-seung’s team at the Pohang Accelerator Laboratory conducted research on the analysis of the coordination structure of single-atom catalysts. Professor Kim Hyung-joon’s team at KAIST specifically identified the cause of changes in catalytic reactivity through theoretical calculations. It was found that this is due to changes in adsorption energy between the catalyst and reaction intermediates depending on the coordination structure.
Professor Joo Sang-hoon of UNIST said, “Understanding the unique properties of single-atom catalysts is important for developing single-atom catalysts, which are core materials for future chemical technologies.” He added, “This study revealed that residual ligands of single-atom catalysts affect catalytic activity and selectivity, giving momentum to the development of single-atom catalysts.”
This research was published online on August 4 in the prestigious chemistry journal ‘Angewandte Chemie International Edition.’
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
The research was supported by the ‘Mid-career Researcher Support Project,’ ‘Future Materials Discovery Project,’ and ‘Climate Change Response Technology Development Project’ promoted by the Ministry of Science and ICT and the National Research Foundation of Korea.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.








