GIST Research Team Develops High-Efficiency Water Electrolysis Catalyst Reducing Iridium Usage by Half
[Asia Economy Honam Reporting Headquarters Reporter Lee Gwan-woo] Gwangju Institute of Science and Technology (GIST) announced on the 25th that the research team led by Professor Park Chan-ho of the Graduate School of Energy Convergence at the Convergence Technology Institute has developed a liquid-phase catalyst synthesis method to produce high-efficiency iridium oxide catalysts applicable to renewable energy-linked water electrolysis.
The research team significantly improved the catalytic activity and stability for the oxygen evolution reaction occurring at the anode of water electrolysis by controlling the oxidation state of iridium.
As a result, the catalyst showed 1.1 times superior activity compared to commercial catalysts and maintained 5.3 times better activity even after stability evaluation.
The polymer electrolyte membrane water electrolysis system, which is easy to link with renewable energy generation, uses iridium-based catalysts, a precious metal, as anode catalysts for the oxygen evolution reaction.
Water electrolysis technology produces hydrogen by electrolyzing water, emitting no carbon dioxide, enabling environmentally friendly green hydrogen production; however, iridium used as a catalyst is a scarce metal resource and expensive, which is a drawback.
Moreover, the current low water electrolysis efficiency is an obstacle to activating green hydrogen production, and active research is underway to improve activity and durability by developing iridium alloys and controlling the oxidation state of iridium.
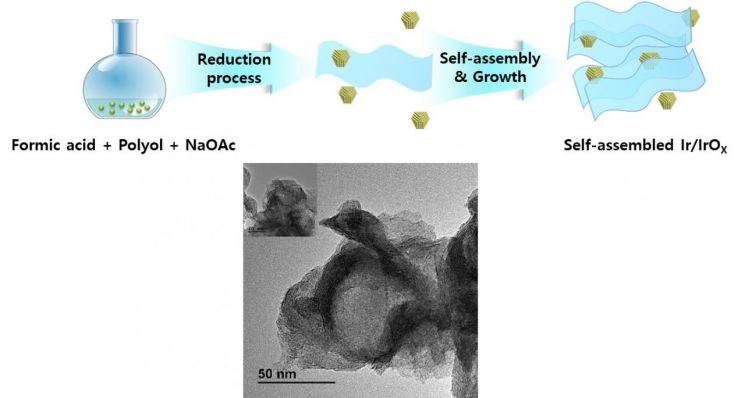
The research team developed a new liquid-phase reduction method to change and optimize the iridium oxidation state on the catalyst surface and core.
When the iridium oxide catalyst developed by the research team was applied to a water electrolysis unit cell, significantly improved performance was confirmed compared to existing commercial catalysts, even though the catalyst usage was less than 50%.
The synthesized catalyst has a gradient structure of oxidation states, with Ir(3+) and Ir(4+) present on the surface and Ir(0) increasing toward the core.
This structure improved both the activity and stability of the oxygen evolution reaction, and when the synthesized catalyst was applied to a water electrolysis unit cell, it showed 5.3 times superior activity compared to commercial catalysts after initial stability evaluation, despite using less than half the amount of iridium catalyst.
Hot Picks Today
![About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]](https://cwcontent.asiae.co.kr/asiaresize/93/2026051416263163580_1778743590.jpg) About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
About 100 Trillion Won at Stake... "Samsung Strike Is an Unprecedented Opportunity" as Prices Surge 20% [Taiwan Chip Column]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- "Envious of Korean Daily Life"...Foreign Tourists Line Up in Central Myeongdong from Early Morning [Reportage]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
Professor Park Chan-ho said, “By using a new liquid-phase reduction method, we created a gradient distribution of iridium oxidation states within the iridium oxide catalyst, simultaneously enhancing activity and durability,” adding, “As demonstrated in the unit cell, it is expected that applying this to actual systems will not only increase water electrolysis efficiency but also reduce the amount of iridium used at the anode in the future.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.