"Unauthorized Raw Material Use" Hans Biomed Artificial Breast Sales Suspension
[Asia Economy Reporter Cho Hyun-ui] On the 13th, the Ministry of Food and Drug Safety confirmed that the medical device company Hans Biomed manufactured and distributed approximately 70,000 units of the silicone gel breast implant "Bellagel" using raw materials different from those approved.
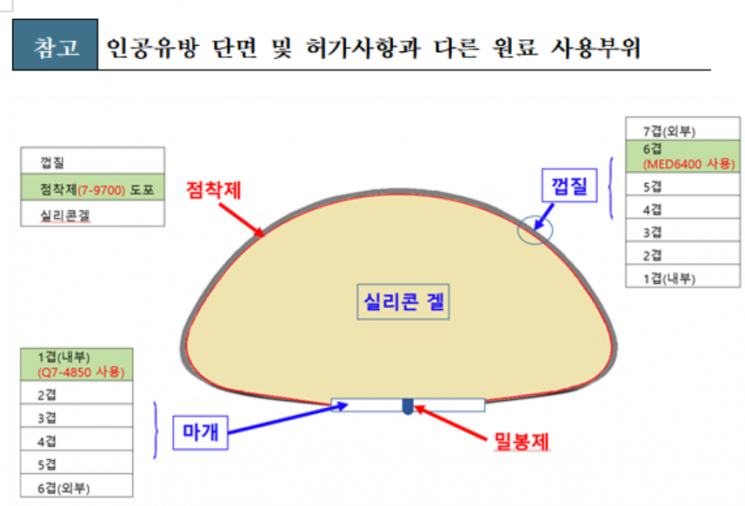
Silicone gel breast implants are products used for breast reconstruction or cosmetic surgery, consisting of silicone gel contained within a silicone pouch.
According to the Ministry's inspection, Hans Biomed produced non-compliant breast implants using unapproved raw materials from December 2015 and supplied about 70,000 units to medical institutions.
There are a total of five types of raw materials not listed in the approval. Among these, the silicone adhesive is used in skin-contact medical devices (wound protectors), and the other four are raw materials used in other domestically approved implantable medical devices (such as breast implants and heart valves).
Experts assessed that since the five unapproved raw materials are mostly used in other implantable medical devices, the possibility of leakage under normal conditions is very low, thus the risk to implant patients is considered minimal. However, the Ministry judged that continuous monitoring of implant patients is necessary.
Additionally, formaldehyde gas, known to potentially occur during the high-temperature manufacturing process of breast implants, is believed by experts to have minimal residual presence in the product after internal air removal processes. The Ministry's residual formaldehyde test on the finished product detected none.
The Ministry ordered the suspension of sales and recall of the product and requested related organizations such as the Society of Plastic Surgery and the Medical Association to instruct medical institutions to stop using the product. Hans Biomed is expected to face administrative sanctions such as business suspension for violating the Medical Device Act.
To ensure the safety and reassurance of patients who received the product, the Ministry will promote ▲ rapid patient identification ▲ provision of safety information ▲ establishment of a dedicated communication channel ▲ long-term monitoring of implant patients ▲ compensation measures.
Currently, the Ministry is identifying patients who underwent breast reconstruction surgery using the product. It plans to provide information on regular examination items, diagnostic procedures, and patient response guidelines to medical institutions for individual implant patients.
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "SK hynix GDRs Trade at a Premium...Global Demand Set to Surge"[Click e-Stock]
- "High-Net-Worth Investors Managing 10 Trillion Won: 'Gangnam Wealthy Also Feel F...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
The Ministry has requested Hans Biomed to prepare and submit compensation plans for implant patients. It plans to promptly finalize the coverage, scope, and duration of compensation for diagnostic and examination costs and adverse effects. Furthermore, it will establish plans for long-term patient monitoring through discussions with experts from various fields and analyze and evaluate the results.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.