MFDS: "White particles found in flu vaccines... 615,000 doses recalled" (Comprehensive)
"Low Safety Concerns Beyond Local Effects"... Only One Reported Adverse Case
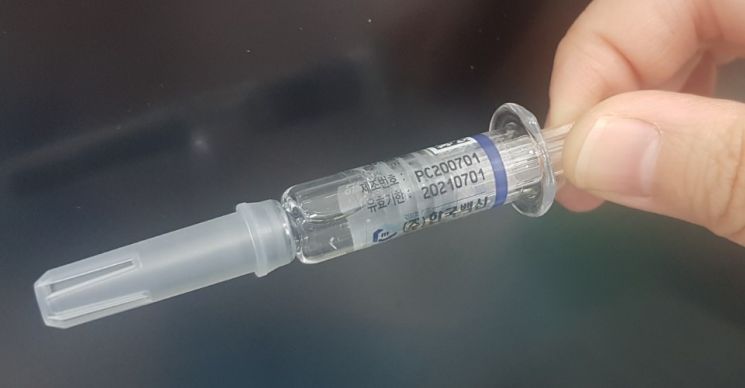
[Asia Economy Reporter Seo So-jeong] White particles were discovered in influenza (flu) vaccines, prompting the manufacturer to voluntarily recall the products.
On the 9th, the Ministry of Food and Drug Safety (MFDS) announced during a briefing at the Korea Disease Control and Prevention Agency that the manufacturer will voluntarily recall a total of 615,000 units from four production batches (PC200701, PC200702, PC200801, PC200802) of Korea Vaccine's influenza vaccine 'Cobaxflu Quadrivalent PF Injection.'
On the 6th, the MFDS received a report from the Yeongdeok-gun Public Health Center in Gyeongbuk Province that white particles were found in the 'Cobaxflu Quadrivalent PF Injection (batch number: PC200701).' The MFDS immediately collected the products for testing. Additionally, they conducted on-site inspections of the manufacturer, analyzed the cold chain (refrigerated distribution), sought expert advice, performed additional product sampling and testing, and requested the manufacturer to submit their own investigation results.
Analysis of the white particles collected during the inspection revealed that particles larger than 75㎛ consisted of 99.7% protein and 0.3% silicone oil. According to expert consultation, the white particles are believed to be due to differences in vaccine components and syringe manufacturing methods, which can cause variations in adsorption and aggregation patterns. It was also understood that physical impacts during distribution and the passage of time could cause the particles to grow larger.
On the day, MFDS Commissioner Lee Eui-kyung stated, "The white particles are considered antigen protein aggregates, and experts believe that aside from local reactions such as pain and inflammation at the injection site, there is a low safety concern." He added, "It is not uncommon for antigen proteins in vaccines to aggregate and form particles."
Commissioner Lee further explained, "Based on on-site inspections of the manufacturer and expert opinions, it is judged that there are no issues with the efficacy and safety of 'Cobaxflu Quadrivalent PF Injection.' However, to reassure the public, the manufacturer has decided to voluntarily recall the influenza vaccines in which white particles were detected."
"Aggregation can occur during vaccine and container manufacturing processes"
Possible due to physical impacts and temperature during distribution
As of the 9th, 17,812 people have been vaccinated with the affected vaccine
On the same day, Professor Choi Won-seok of the Department of Infectious Diseases at Korea University Ansan Hospital said, "Aggregation can occur due to the concentration of components used in the vaccine or during the manufacturing processes of the vaccine or container, including the use of drugs, acids, or silicone treatment. Physical impacts and temperature conditions during distribution can also cause white substances to appear over time."
Regarding the effects on the human body, Professor Choi stated, "If the white particles are indeed antigen protein aggregates composed of proteins and antigens, the likelihood of local reactions such as pain, redness, or swelling at the injection site may increase. However, experts believe that the possibility of systemic adverse reactions is low."
He added, "There were some differing opinions regarding efficacy, but if there is no difference in antigen quantity, it is presumed that there will be no difference in efficacy."
According to the Korea Disease Control and Prevention Agency's integrated vaccination management system, as of 3 p.m. on the 9th, 17,812 people have been vaccinated with the four production batches of Cobaxflu Quadrivalent PF Injection (7,018 recipients under the National Immunization Program and 10,794 general paid recipients). Only one adverse event (local pain) has been reported so far.
The MFDS is cooperating with the Korea Disease Control and Prevention Agency to promptly inform medical institutions supplied with the affected batches about the recall and is urging medical institutions to suspend use of the product and actively cooperate with the manufacturer's recall.
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- Chairman Seungyeon Kim of Hanwha Group Encourages Planners: "Heroes and Pride"
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
Commissioner Lee emphasized, "Before using the influenza vaccine, it should be shaken sufficiently, and if discoloration or sedimentation is observed with the naked eye, it should not be used. We will establish improvement measures such as strengthening verification of test items related to white particles at the national lot release stage and strive to prevent such issues from occurring."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.