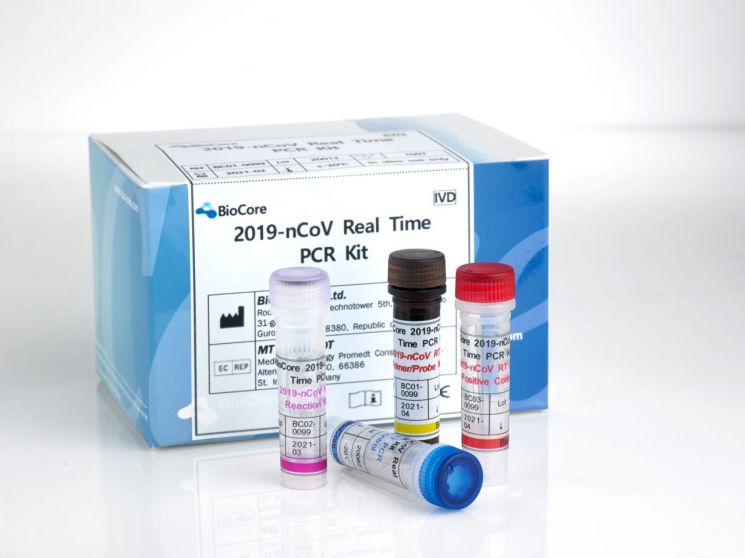
BioCore COVID-19 Diagnostic Kit, Sixth Emergency Approval in Korea
[Asia Economy Reporter Choi Dae-yeol] BioCore, a molecular diagnostic specialized bio company, received emergency use authorization from the Korea Disease Control and Prevention Agency on the 11th for its novel coronavirus infection (COVID-19) diagnostic kit. It is the sixth domestic diagnostic kit to be approved.
The approved BioCore product (BioCore 2019-nCoV Real Time PCR Kit) had previously obtained export approval from the Ministry of Food and Drug Safety and CE certification in Europe. The company explained that the kit uses real-time RT-PCR method and showed 100% sensitivity and specificity in its own evaluation, indicating high reliability.
Exports are underway to more than 20 countries worldwide, including European regions such as Germany, Italy, and Spain, as well as India, Saudi Arabia, and Ghana. The company has also applied for emergency use authorization from the U.S. Food and Drug Administration (FDA) and expects approval in the near future.
The Korea Disease Control and Prevention Agency had previously received applications around the end of January and approved five products after performance testing. These are diagnostic kits from CoGen Biotech, Seegene, SolGent, SD Biosensor, and BioSewoom. All are in vitro diagnostic devices using patient specimens with the RT-PCR method.
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- "Target Price Set at 970,000 Won"... Top Investors Already Watching, Only an 'Uptrend' Remains [Weekend Money]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.