The "Soil" Changes Before Cancer Emerges... GIST Identifies Pre-Developmental Stage of Lung Cancer [Reading Science]
Mutated Cells Reshape Surrounding Tissue
A New Therapeutic Strategy to Block Cancer at Its Onset
For the first time, it has been revealed that mutated cells transform their surrounding environment into “fertile ground” for cancer growth long before lung cancer develops into a visible tumor. This finding suggests that advancing beyond conventional treatments that remove cancer after it is detected, it may be possible to develop preventive therapeutic strategies that block the onset of the disease itself.
On April 29, the Gwangju Institute of Science and Technology (GIST) announced that a research team led by Professor Jinwook Choi from the Department of Life Sciences, in collaboration with Professor Juhyun Lee’s team from Memorial Sloan Kettering Cancer Center (MSK), has, for the first time in the world, identified the “cascade” structure of intercellular reactions occurring at the earliest stages of lung cancer. The results of this research were published online in the international journal Nature on April 22.
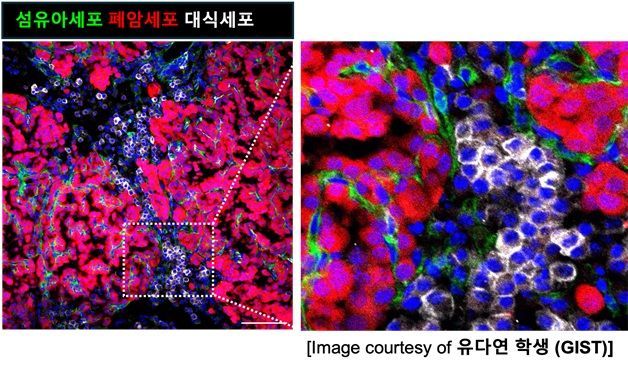
It shows the process in early lung cancer where mutant stem cells send signals to surrounding fibroblasts and macrophages, transforming the tissue environment into a cancer-friendly one and promoting tumor growth. Provided by the research team
View original imageLung adenocarcinoma (LUAD) is known for its high mortality rate, but is often only detected at an advanced stage due to a lack of early symptoms. While it has been known that KRAS mutations occurring in alveolar type 2 cells (AT2) can lead to cancer, the process by which these mutated cells transform the surrounding tissue into a “cancer-friendly environment” had not been clarified until now.
'Mutation → Fibrosis → Inflammation': The Three-Step Circuit That Fuels Cancer
The research team tracked intercellular interactions during the early stages of cancer development using mouse models and 3D lung organoid experiments. They discovered that mutated lung stem cells recruit neighboring cells to form a “self-amplifying circuit” that facilitates tumor formation.
In the first stage, the mutated cells secrete large amounts of “amphiregulin (AREG),” sending signals to nearby cells. In the second stage, fibroblasts that receive this signal lose their tissue repair function and transition into a “fibrotic state” that hardens the tissue.
In the third stage, this altered environment attracts immune cells, intensifies inflammatory responses, and completes a vicious cycle that once again promotes cancer cell growth. Ultimately, this creates a “soil-seed” structure in which the cancer cells and their environment support each other, leading to the full development of tumors.

Research team photo. (From left) Professor Jinwook Choi of Gwangju Institute of Science and Technology (GIST) Department of Life Sciences, PhD candidate Hyeyoung Lee, (top right from left) PhD candidate Eric Cardoso and Professor Juhyun Lee from Memorial Sloan Kettering Cancer Center (MSK). Provided by GIST
View original imageBlocking Signals Suppresses Cancer Development: "A Turning Point for Preventive Therapy"
The research team also found that genetically or pharmacologically blocking the amphiregulin signaling pathway—the core of this process—suppressed the formation of the fibrotic microenvironment and significantly reduced the early development of lung cancer. This suggests a potential therapeutic target for intervention not after cancer appears, but before it even develops.
In collaboration with Park Moosuk’s team at Severance Hospital, Yonsei University, the researchers confirmed that the same phenomena occurred in organoid models simulating the patient environment. These results support the possibility that this mechanism operates in actual clinical settings as well.
Hot Picks Today
![Home Appliance Woes Mount, Yet 45 Trillion Won in Bonuses? Samsung's Risky Asymmetry [Why&Next]](https://cwcontent.asiae.co.kr/asiaresize/93/2022111609212518207_1668558086.jpg) Home Appliance Woes Mount, Yet 45 Trillion Won ...
Home Appliance Woes Mount, Yet 45 Trillion Won ...
- "Major Crash Is Coming... Buy Even If You Have to Skip a Meal" 'Rich Dad' Shares...
- Government Accelerates Creation of Korean Sovereign Wealth Fund... Establishes F...
- 500% Energy Efficiency... Samsung Electronics’ 'Energy Magic' to End the Fear o...
- "It Was Fantastic" Jensen Huang's Daughter Seals 'Robot Alliance' with LG throug...
Professor Jinwook Choi stated, “We have proposed a strategy that fundamentally blocks the onset of cancer by disrupting the ‘dialogue’ between cancer cells and their environment, moving beyond the conventional approach of targeting cancer cells alone. This could mark a paradigm shift towards preventive and precision therapies that suppress lung cancer at its earliest stage.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.