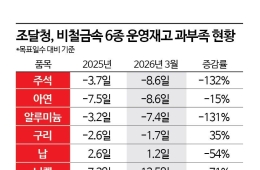
Kolon TissueGene Unveils 17-Year Long-Term TG-C Data at OARSI
Zero Cases of Tumor Development Confirmed, Efficacy Also Observed in Obese Patient Group
Topline Results from Phase 3 Trial Expected in July
Kolon TissueGene has released long-term safety data for TG-C, its cell and gene therapy for knee osteoarthritis, accumulated over 17 years, at a leading osteoarthritis conference in the United States. This disclosure comes approximately three months ahead of the announcement of the topline results from its Phase 3 clinical trial.

A Kolon TissueGene representative is presenting the poster exhibited at the Osteoarthritis Research Society International (OARSI) Annual Meeting held in West Palm Beach, Florida, USA, from the 23rd to the 26th. Kolon TissueGene
View original imageOn April 27, Kolon TissueGene announced that it presented long-term follow-up data for TG-C, along with efficacy analysis results by body mass index (BMI) from its Phase 2 clinical trial, through a poster session at the Osteoarthritis Research Society International (OARSI) Annual Meeting held from the 23rd to the 26th in West Palm Beach, Florida, USA.
The long-term follow-up data covers observations of 219 participants over approximately 17 years as of July 31, 2025. During this period, there were no reported cases of tumor development associated with TG-C. Furthermore, no safety concerns were identified in a subgroup of 30 elderly patients who were followed for more than 10 years.
The efficacy analysis by BMI revealed an even more pronounced pain improvement in obese patients (BMI of 30 or higher). Based on the Phase 2 trial data, the improvement in pain scores (VAS) compared to the placebo group was -18.4 in the group with BMI under 30, and -26.1 in the group with BMI 30 or higher. The company plans to conduct an analysis of the Phase 3 data and officially announce the first topline results in July.
Hot Picks Today
![Silently Climbing to the Top... Will Samsung Electronics Become the World's Most Profitable Company? [Why&Next]](https://cwcontent.asiae.co.kr/asiaresize/93/2026042710274741455_1777253267.png) Silently Climbing to the Top... Will Samsung El...
Silently Climbing to the Top... Will Samsung El...
- If You Had Followed, You'd Have Doubled Your Money... Presidential Fund Returns ...
- "3,000 Cups of Coffee Despite Zero Revenue"... O-world Caf? Owner Supports Neukg...
- "I Want to Sleep Here"... The Reason a Girl Lay Beside Her Grandparents' Grave a...
- Lingering at the Olive Young Shelf, Then Straight Into the Basket... "Not Cosmet...
Moonjong Noh, CEO of Kolon TissueGene, stated, "The data accumulated over 17 years provides strong evidence supporting the safety of TG-C," adding, "Consistent efficacy has been demonstrated regardless of obesity status." Seungho Jeon, CEO of Kolon TissueGene, commented, "TG-C is the only therapy for which we have secured long-term data of over 15 years from real-world clinical cases," and added, "We are preparing strategies to leverage this data in the commercialization phase."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.