AppClon Partner Henlius Unveils HER2 ADC at AACR, Shows Antitumor Activity Superior to Enhertu
"Complete Remission Observed in HER2 Ultra-Low Expression Models,
Excellent Safety Profile Confirmed Even at High Doses"
AppClon, a company specializing in antibody-based novel drug development, announced on April 22 that its global partner, Henlius of China, presented preclinical data on its next-generation antibody-drug conjugate (ADC) candidate 'HLX49' at the American Association for Cancer Research (AACR) 2026 held in San Diego, USA.
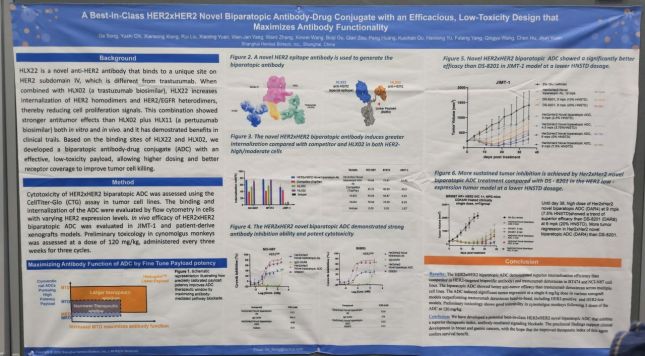
Poster of Abclon displayed at the American Association for Cancer Research (AACR) 2026 held in San Diego, USA. Abclon
View original imageHLX49 is a HER2-targeted ADC based on AppClon's antibody technology AC101 (HLX22). It features a bispecific paratopic structure that can simultaneously recognize different binding sites. According to the company, this induces HER2 receptor internalization and increases drug delivery efficiency.
According to materials released by Henlius, HLX49 demonstrated higher internalization efficiency and antitumor activity compared to trastuzumab deruxtecan (T-DXd, Enhertu) in breast cancer (BT-474) and gastric cancer (NCI-N87) cell line experiments. In animal studies, a single dose of 6 mg/kg resulted in tumor reduction, and antitumor activity was also confirmed in HER2 ultra-low expression models (IHC less than 1+). Regarding safety, preliminary toxicity tests in primates showed no significant toxicity even at repeated doses of up to 60 mg/kg.
Henlius aims to submit an Investigational New Drug (IND) application for HLX49 within this year.
Hot Picks Today
![[Exclusive] Raw Pork Lard Illegally Distributed as Edible Ingredient, Supplied Even to Department Stores](https://cwcontent.asiae.co.kr/asiaresize/93/2026042109395833880_1776731998.jpg) [Exclusive] Raw Pork Lard Illegally Distributed...
[Exclusive] Raw Pork Lard Illegally Distributed...
- Apartment or Theme Park? From Protruding Terraces to Unmanned Shuttles... Altern...
- Even With a 900 Million Won Deposit and 2.5 Million Won Monthly Living Expenses,...
- Seoul Metropolitan Area Apartment Presale Prices Rise 16.6% in One Year
- Detained Jung Yura Appeals from Prison: "My Children Will Be Sent to an Orphanag...
The base antibody for HLX49, AC101 (HLX22), is currently undergoing clinical trials for six indications including gastric cancer. Among these, a global phase 3 trial for first-line treatment of metastatic gastric cancer is underway, and patient enrollment has been completed for a phase 2 combination trial in HER2 low-expression patients.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.