Busan Paik Hospital Begins Full-Scale Prescription of Alzheimer’s New Drug ‘Leqembi’
Inje University Busan Paik Hospital (Director Lee Yeon-jae) is set to begin full-scale treatment by introducing the new drug for mild Alzheimer's disease, "Leqembi."
Busan Paik Hospital announced on the 29th that Professors Kim Sang-jin, Jung Eun-joo, and Jung Jin-ho from the Department of Neurology will start prescribing Leqembi (generic name: lecanemab) from January next year for patients with mild cognitive impairment and mild Alzheimer's disease.
Leqembi is an Alzheimer's treatment jointly developed by the American company Biogen and the Japanese company Eisai. It received FDA approval in the United States in July 2023, was approved by the Ministry of Food and Drug Safety this year, and was introduced domestically for the first time starting this month.
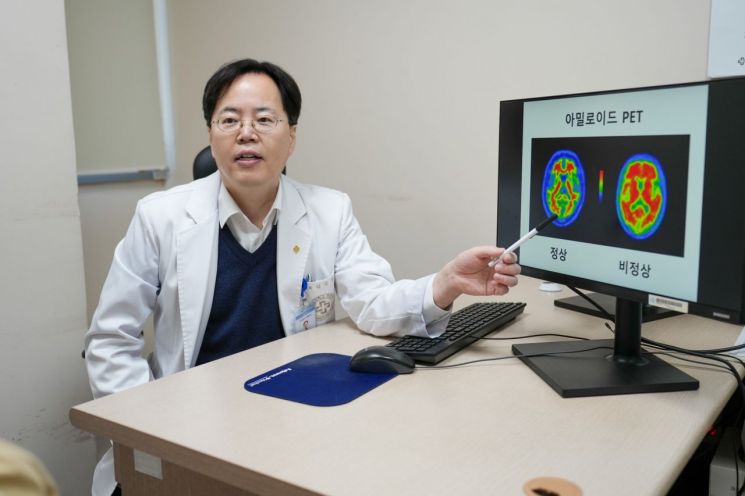
According to Paik Hospital, one of the causative substances of Alzheimer's disease, "amyloid beta," begins to accumulate in the brain about 20 years before the onset of the disease. When amyloid beta accumulates in the brain, it causes brain cell damage and triggers Alzheimer's disease, leading to various symptoms such as memory loss and cognitive decline, which gradually worsen.
Leqembi selectively removes the highly toxic amyloid beta, slowing brain damage and delaying the progression of Alzheimer's disease symptoms. Clinical studies have shown that patients treated with Leqembi for 18 months experienced approximately a 78% reduction in amyloid beta accumulation and about a 27% delay in symptom progression.
However, the efficacy and safety for patients with moderate or more advanced Alzheimer's disease have not been confirmed. The treatment is administered intravenously every two weeks over about one hour. Additionally, regular MRI scans are required during treatment to monitor for adverse reactions such as brain edema and microbleeds.
Professor Kim Sang-jin of Neurology explained, "Before prescribing Leqembi, Alzheimer's disease diagnosis and suitability are determined through cognitive function tests (neuropsychological tests), brain MRI scans, amyloid PET scans, blood tests, and APOE genetic testing. Until now, Alzheimer's treatments have not eliminated the fundamental cause of the disease but have been used to alleviate some dementia symptoms."
Hot Picks Today
 "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
"Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Already Watching Closely..."Target Price Set at 970,000 Won" Only Upward Momentum Remains [Weekend Money]
- [Breaking] Prime Minister Kim Minseok Issues Public Statement on Samsung Electronics Strike: "Welcoming Resumption of Negotiations... Effectively the Last Opportunity"
- Did Samsung and SK hynix Rise Too Much?... Foreign Assets Grow Despite Selling [Weekend Money]
- Is It Really Like an Illness? "I Can't Wait to Go Again"—Over 1 Million Visited in Q1, Now 'Busanbyeong' Takes Hold [K-Holic]
Professor Kim added, "With the emergence of this new drug that slows disease progression after about 20 years, patients with mild symptoms can delay progression to moderate and severe stages. This is expected to significantly contribute to reducing the social costs involved in diagnosing, treating, and caring for dementia patients."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.