Celltrion Achieves Highest Ever 1Q Sales... Led by Remsima
Celltrion recorded its highest-ever sales in the first quarter, driven by the growth of its autoimmune disease treatment ‘Remsima’.
Celltrion announced on the 8th that its consolidated sales for the first quarter of this year were tentatively estimated at 597.5 billion KRW, with an operating profit of 182.3 billion KRW. Compared to the same period last year, sales increased by 12.4% and operating profit rose by 41%. The operating profit margin during this period was 30.5%.
The company attributed the increase in first-quarter sales to the growth in biopharmaceutical sales. Celltrion’s biopharmaceutical sales in the first quarter of this year reportedly increased by 49% compared to the same period last year. Biosimilars also surpassed 400 billion KRW in quarterly sales for the first time ever.
The increased sales proportion of the highly profitable RemsimaSC positively contributed to improved profitability. Remsima (active ingredient infliximab) is an antibody biosimilar developed and produced by Celltrion, and RemsimaSC is a formulation of this product in a subcutaneous (SC) injection form. Notably, RemsimaSC is the world’s first and only case of an infliximab product developed and launched as a subcutaneous injection.
The Remsima product line is expanding its market share primarily in the European market. As of the fourth quarter of last year, Remsima and RemsimaSC held a 60.6% market share in Europe, which is higher than that of the original drug ‘Remicade.’ RemsimaSC recorded a 16.1% market share in the five major European countries. Specifically, it holds a 32% market share in Germany and 21% in France. The combined market share of Remsima and RemsimaSC in these markets reaches 67.9%. In the U.S. market, Remsima (marketed as Inflectra in the U.S.) held a 31.4% market share as of the first quarter.
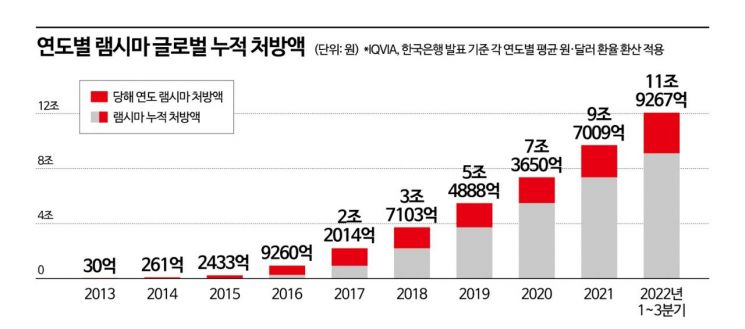
Remsima holds the record for the highest cumulative prescription amount among pharmaceuticals developed and sold by domestic pharmaceutical and bio companies. According to IQVIA, a pharmaceutical market research firm, Remsima recorded a cumulative global prescription amount of 11.9267 trillion KRW from 2013 through the third quarter of last year. By the end of last year, the prescription amount surpassed 12 trillion KRW. During this period, the global cumulative sales amounted to 5.1631 trillion KRW. Remsima is the first single product among domestically developed and produced pharmaceuticals to exceed 12 trillion KRW in global cumulative prescriptions and 5 trillion KRW in cumulative sales.
There is also potential for expansion in the U.S., the world’s largest pharmaceutical market. RemsimaSC is currently undergoing the approval process as a new drug in the U.S., and Celltrion expects to obtain U.S. Food and Drug Administration (FDA) approval for RemsimaSC around October. Although Remsima was originally developed as a biosimilar, the FDA highly regarded the potential of RemsimaSC and recommended applying for new drug approval. The new drug application was submitted at the end of last year, and the approval process is currently underway.
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
Experts have assessed that Remsima’s growth, led by market expansion, will have a positive impact on Celltrion’s profitability. Hyunsoo Ha, a researcher at Yuanta Securities, analyzed, “Unlike the Humira market, where price competition is intense, RemsimaSC is the only subcutaneous injection formulation in the Remicade market. Along with the convenience of self-administration and the elimination of hospital visit costs, it has an advantage in price competition, and after its expected U.S. launch in 2024, it is projected to contribute significantly to profits.”
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Remsima SC. [Image provided by Celltrion]](https://cphoto.asiae.co.kr/listimglink/1/2020060410565875880_1591235818.jpg)