UNIST Develops Catalyst Design Method for Easier Conversion of Toxic Substances
[Asia Economy Yeongnam Reporting Headquarters Reporter Hwang Dooyul] A catalytic design method has been developed that can easily oxidize toxic substances emitted from industrial and living environments using ‘metal-reactive oxygen species’ formed by the interaction of metal enzymes and reactive oxygen species present in the human body.
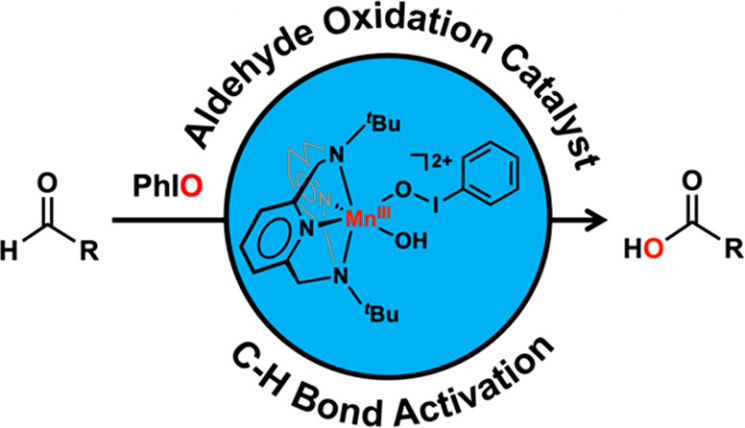
The research team led by Professor Jaeheung Cho of the Department of Chemistry at UNIST identified that one type of metal-reactive oxygen species, the ‘manganese-iodosylbenzene species,’ oxidizes toxic ‘aldehyde compounds’ into ‘carboxylic acids,’ and that this reaction follows a novel electrophilic mechanism.
Aldehydes are byproducts in the petroleum industry or occur in daily life. When mixed in the air, these substances cause air pollution and are known carcinogens that induce mutations by exhibiting genotoxicity within living organisms. They are formed as byproducts during the metabolic process of alcohol decomposition in the body and have significant effects. Therefore, decomposing these substances is recognized as important in the fields of environmental chemistry and biochemistry.
To oxidize aldehydes, strong oxidizers such as potassium permanganate (KMnO4) or strong basic conditions to induce disproportionation reactions are required. Additionally, nucleophiles are needed to initiate the reaction.
In this study, a metal catalyst was developed that can convert aldehydes into carboxylic acids under mild conditions without the aid of nucleophiles.
Donghyun Jeong, the first author and a researcher in the Department of Chemistry at UNIST, said, “By designing a catalyst that can oxidize aldehydes stably through an electrophilic attack different from conventional methods, we were able to convert aldehydes into carboxylic acids even under mild conditions.”
In nature, metal enzymes form ‘metal-reactive oxygen intermediates’ using external oxygen and electrons, and these intermediates directly participate in the oxidation reactions of organic substances.
In the study, the team synthesized the ‘manganese-iodosylbenzene species’ using iodosylbenzene and elucidated the process by which this species participates in the oxidation reaction of aldehydes. They also proposed a new aldehyde oxidation reaction system through kinetic studies.
Professor Jaeheung Cho of the Department of Chemistry stated, “It is academically significant that we developed a new aldehyde oxidation catalyst utilizing the proton absorption property of the biomimetic synthesized ‘manganese-iodosylbenzene species.’” He added, “This research will help future studies on developing catalysts that decompose environmental pollutants in environmental and industrial fields and on converting aldehydes into various functional groups.”
The research was jointly led by first authors Donghyun Jeong, a researcher, and Hyokyung Kim, a doctoral student, from Professor Cho’s research team. The results were published on the 4th in the international chemistry journal ‘Journal of the American Chemical Society (JACS).’
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
The research was supported by the Basic Research Project (Mid-career Researcher) of the Ministry of Science and ICT and the National Research Foundation of Korea, as well as the Step Leap Carbon Neutral Technology Development Project.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.