Finally, the 'Oral Treatment' Arrives... Will It Be a 'Game Changer' Effective Against Variants?
[Asia Economy Reporter Lee Chun-hee] Oral COVID-19 treatments are expected to arrive as early as this week. These treatments are anticipated to significantly reduce the likelihood of hospitalization when taken early, and they are also expected to maintain their effectiveness against the continuously emerging COVID-19 variants, raising public expectations.
According to the government on the 9th, detailed plans for the introduction and utilization of oral treatments will be separately announced within this week. The first batch of these treatments is also expected to arrive domestically as early as this week.
Currently, the government has secured a total of 1,004,000 doses of oral treatments. This includes 762,000 doses of Pfizer's 'Paxlovid', which has received emergency use authorization from the Ministry of Food and Drug Safety, and 242,000 courses of Merck (MSD)'s 'Molnupiravir'.
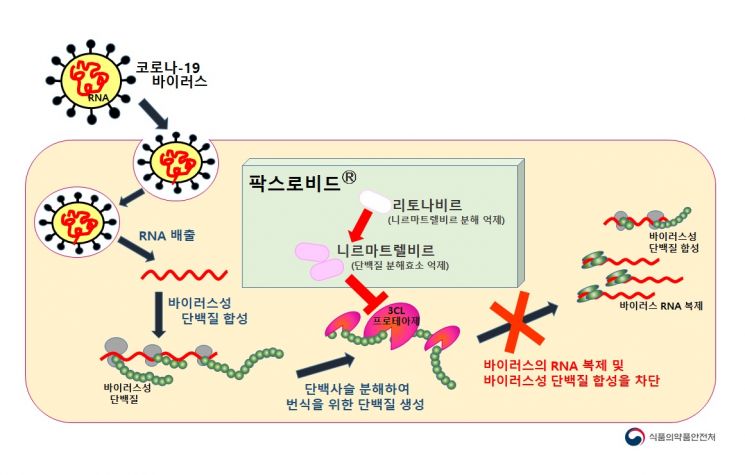
Considering that Paxlovid has already obtained emergency use authorization in Korea, the first batch is likely to be Paxlovid. Paxlovid is taken as a total of three pills simultaneously per dose, consisting of two types of pills. It contains 300 mg of Nirmatrelvir (two 150 mg pills) and 100 mg of Ritonavir (one 100 mg pill), taken twice daily every 12 hours. One course consists of 30 pills, taken as six pills per day for five days. Administration must begin within five days after testing positive for COVID-19 and symptom onset.
The target users of Paxlovid are adults with mild to moderate COVID-19 who are at high risk of progressing to severe disease, as well as pediatric patients aged 12 years and older weighing at least 40 kg. Pregnant women may receive the treatment if the benefits outweigh the risks, and breastfeeding mothers should stop breastfeeding during the treatment period.
One of Paxlovid's greatest strengths is its expected strong efficacy against the continuously emerging COVID-19 variants. In a clinical trial involving 2,246 high-risk non-hospitalized patients with mild to moderate symptoms, administration within five days of symptom onset reduced hospitalization and death rates by 88%, with 98% of the patients infected with the Delta variant.
Unlike existing vaccines or antibody treatments that inhibit viral replication by binding to the spike protein of the COVID-19 virus, Pfizer's Paxlovid blocks the protease enzyme (3CL protease), preventing the production of proteins necessary for viral replication and thereby inhibiting viral proliferation. Since viral mutations mainly occur in the spike protein, it is expected that Paxlovid will maintain its efficacy against most variants.
Hot Picks Today
 Taking Annual Leave and Adding "Strike" to Profiles, "It Feels Like Samsung Has Collapsed"... Unsettled Internal Atmosphere
Taking Annual Leave and Adding "Strike" to Profiles, "It Feels Like Samsung Has Collapsed"... Unsettled Internal Atmosphere
- There Is a Distinct Age When Physical Abilities Decline Rapidly... From What Age Do Strength and Endurance Drop?
- "One Comment Could Lead to a Report": 86% of Elementary Teachers Feel Anxious; Half Consider Resignation or Career Change
- "After Vowing to Become No. 1 Globally, Sudden Policy Brake Puts Companies’ Massive Investments at Risk"
- On Teacher's Day, a Student's Gifted Cake Had to Be Cut into 32 Pieces... Why?
Side effects have also been reported to be mild. Major side effects observed during clinical trials included taste disturbances, diarrhea, increased blood pressure, and muscle pain, but most were mild.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![Pfizer's oral COVID-19 treatment 'Paxlovid' <br>[Image source=Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2021122318215073729_1640251309.jpg)