Another Unauthorized Manufacturing... Samsung Pharmaceutical Suspends Production and Sales of 6 Items
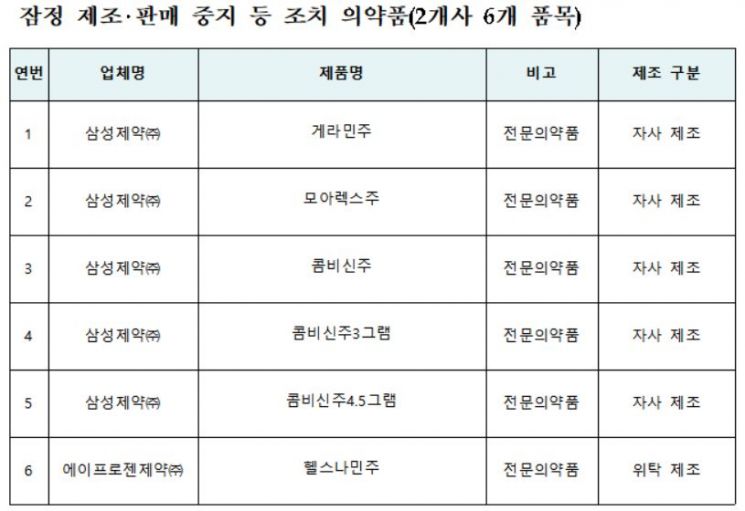
Medicines Subject to Provisional Manufacturing and Sales Suspension Measures (Data from MFDS)
View original image[Asia Economy Reporter Kim Ji-hee] The Ministry of Food and Drug Safety announced on the 8th that it will temporarily suspend the manufacturing and sales of six products, including 'Geraminju' manufactured by Samsung Pharmaceutical, and initiate a recall.
This measure follows a special inspection conducted by the 'Pharmaceutical GMP Special Inspection Team' on Samsung Pharmaceutical, which confirmed violations of the Pharmaceutical Affairs Act, including the unauthorized use of additives without obtaining change approval (notification) and falsification of manufacturing records.
The Ministry of Food and Drug Safety has issued a safety alert requesting cooperation from experts such as pharmacists to switch the six products to alternative medicines and ensure proper product recall. The Ministry of Health and Welfare and the Health Insurance Review and Assessment Service have been asked to take measures to prevent hospitals and clinics from prescribing these products.
Hot Picks Today
 No Bacteria Detected in Arisu After 24 Hours of Repeated Drinking from a Tumbler
No Bacteria Detected in Arisu After 24 Hours of Repeated Drinking from a Tumbler
- "Not Even Expecting Gratitude, Just Hoping for No Complaints": Teachers' Sad Portrait on the Eve of Teacher's Day
- "Available Only in Korea": Pokémon Card Prices Surge 2,532% Due to Rarity, Becoming Investment Assets
- [US-China Summit] Xi Jinping: "China and the US Must Overcome the Thucydides Trap" (Comprehensive)
- "Egg Prices Are Strange" Proven True... Collusive Margin Hike of 46% Without Basis Leads to 594 Million Won Fine
The Ministry stated, "We will continue to operate the special inspection team to conduct unannounced inspections of pharmaceutical manufacturing sites throughout the year, prioritizing patient safety and promptly taking necessary actions."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







