Development of Semiconductor Technology to Prevent Electric Vehicle Battery Fires
Dr. Lee Jung-gi's Team at KIST Develops Technology to Block Causative Substances by Forming Semiconductor Thin Film on Lithium Metal Electrode Surface
[Asia Economy Reporter Kim Bong-su] As the era of electric vehicles approaches, concerns over frequent battery explosions and fire accidents continue unabated. In this context, a domestic research team has developed a technology that drastically reduces the risk of fire and explosion by applying semiconductor technology.
The Korea Institute of Science and Technology (KIST) announced on the 27th that Dr. Lee Joong-ki's research team at the Energy Storage Research Division developed a technology that forms a semiconductor thin film on the surface of lithium metal electrodes to fundamentally block the formation of dendrites, which are the main cause of battery fires.
It is known that dendrites formed on the surface of materials are the biggest cause of fires in lithium-ion secondary batteries. During battery charging, lithium ions move to the anode and are stored as lithium metal on the surface. The dendrites are branch-like crystals formed in this process, which cause electrode volume expansion and reactions between the electrode and electrolyte, leading to fires and degradation of battery performance.
The research team exposed fullerene (C60), a highly conductive semiconductor material, to plasma to create a semiconductor thin film between the lithium metal electrode and the electrolyte, preventing dendrite formation. The developed semiconductor thin film allows electrons to pass but blocks lithium ions. Since electrons and ions cannot meet on the electrode surface, lithium crystals do not form, fundamentally preventing dendrite formation.
To test electrode stability, experiments were conducted using lithium-lithium symmetric cells. While conventional lithium metal electrodes remained stable up to 20 charge-discharge cycles under extreme electrochemical conditions, electrodes with the developed semiconductor thin film remained stable for 1,200 cycles without lithium dendrite growth. Additionally, when evaluating stability using lithium cobalt oxide cathodes and the developed electrodes, about 81% of capacity was maintained after 500 cycles, which is approximately a 60% improvement compared to about 52% capacity retention in conventional lithium metal electrodes.
Dr. Lee Joong-ki of KIST stated, "The high-safety lithium metal electrode technology developed in this study is expected to be recognized as a next-generation convergent foundational technology for developing safe next-generation secondary batteries without fire risks by suppressing metal dendrite formation that occurs in conventional lithium metal." He added, "We plan to conduct research to apply this technology using other inexpensive materials instead of the costly fullerene used to form the semiconductor thin film. By reducing material and processing costs, we aim to move one step closer to commercialization." This research was published in the latest issue of the international materials science journal ‘ACS Energy Letters’ (IF: 19.003, JCR top 1.852%).
Below is the background and content of the research as explained by the research team.
▲Research Background
With increasing dependence on lithium-ion batteries as power sources for various electronic devices including electric vehicles, research on all-solid-state batteries using lithium metal itself as the anode to maximize energy density has been actively conducted recently.
However, barriers due to high costs and infrastructure still exist. Even if all-solid-state separators are introduced, concerns about the instability inherent to lithium metal remain. Under extreme electrochemical reaction environments, irreversible capacity increase, dendrite structures, and dead lithium formation caused by uneven deposition behavior of lithium metal lead to rapid efficiency decline and instability. These issues, inherent to lithium metal properties, have been significant barriers to manufacturing safe lithium electrodes.
▲Research Content
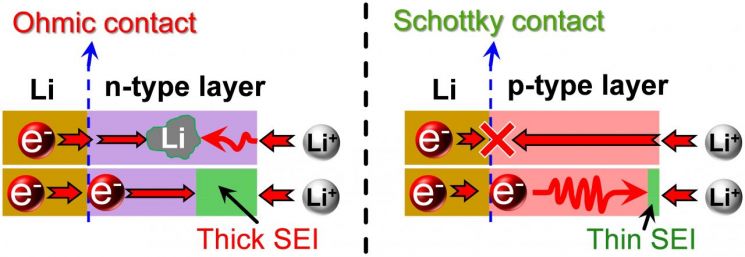
Dr. Lee Joong-ki’s team developed a technology to control the carbon film characteristics derived from fullerene to produce n-type and p-type semiconductors, thereby changing the interfacial properties in contact with lithium metal films to control electrochemical characteristics, aiming to improve structural stability and suppress dendrite formation in lithium metal. When a semiconductor plasma carbon film contacts lithium metal, an Ohmic contact interface forms with n-type semiconductor films. In this case, electrons and ions pass through the semiconductor film, depositing lithium in the semiconductor layer, resulting in unstable lithium electrodes. Conversely, when a p-type carbon film contacts lithium, a Schottky contact characteristic forms. The semiconductor layer blocks electrons but allows ions to pass, causing lithium metal to deposit only beneath the semiconductor layer. This not only prevents dendrite formation but also significantly reduces irreversible capacity, enabling the manufacture of electrochemically stable lithium metal electrodes.
The electrodes developed by the research team, featuring lithium metal and plasma polymerized carbon films with Schottky interfaces, showed 60 times improved cycle stability compared to conventional lithium metal electrodes without artificial interfaces, even under extreme electrochemical conditions of 10 mA/cm² current density and 10 mAh/cm² current capacity, without lithium dendrite growth. The artificial interface with p-type plasma polymerized carbon film exhibiting Schottky contact with lithium possesses semiconductor properties characterized by a majority hole carrier nature, resulting in high ionic conductivity and low electronic conductivity. Due to the structural and electrochemical stability of these lithium metal electrodes, safety evaluations using lithium cobalt oxide as the counter electrode showed about 60% improved capacity retention over 500 cycles compared to conventional lithium electrodes under a current density of 1C.
Below is a Q&A with the research team.
▲What motivated or was the background for starting this research?
Lithium metal, which has high capacity, continuously reacts with electrolytes, causing unstable SEI (Solid Electrolyte Interphase) and dendrite formation, leading to instability in electrochemical environments and difficulties in practical application. Although research on all-solid-state batteries using solid separators is actively underway, barriers due to high costs and infrastructure remain, and concerns about instability inherent to lithium metal persist even if solid separators are introduced.
▲What is different about this achievement?
This study developed a foundational technology that applies semiconductor physics concepts for the first time to improve metal electrodes in secondary batteries. By controlling carbon film characteristics derived from fullerene to produce n-type and p-type semiconductors and changing the interfacial properties in contact with lithium metal films, the technology enables lithium ions to be reduced and deposited only in specific areas where dendrite formation does not occur. This basic technology can be applied to manufacture high-safety electrodes for multivalent metals, which still face application challenges.
▲How could this be utilized if commercialized?
The currently developed technology is more significant as an academic convergence technology implementation rather than an immediately commercializable technology. It is meaningful as a method to manufacture high-safety lithium electrodes by combining theoretical and experimental approaches to secondary battery interfacial phenomena using semiconductor physics theory.
▲What are the expected effects and challenges for commercialization?
The development of high-safety lithium metal electrodes is expected to be used as energy sources for the 4th industrial revolution, including electric vehicles, wearable robots, and drones. For commercialization, improvements in affordable precursors and coating technologies are necessary.
Below are explanations of key terms.
▲Dendrite
A lithium metal crystal formed in a branch-like shape due to electrons concentrating at uneven tips on the lithium metal surface during lithium ion deposition and removal. Continuous dendritic growth causes electrolyte depletion, gas generation, increased internal battery pressure, safety issues, and reduced battery lifespan.
▲1C rate
The rate of charging and discharging a battery fully (100% capacity) in one hour.
▲SEI (Solid Electrolyte Interphase)
Hot Picks Today
 "Do We Need to Panic Buy Again?" War Drives 30% Price Surge... Even the Bedroom Feels the Impact
"Do We Need to Panic Buy Again?" War Drives 30% Price Surge... Even the Bedroom Feels the Impact
- [Breaking] Lee Jae-yong: "All Samsung members are united as one... We must pool our wisdom and move in a single direction"
- "Is a 10,000 KOSPI Breakthrough Possible?" Target Index Raised by 40%... Securities Firms Release Outlook [Weekend Money]
- "Anyone Who Visited the Room Salon, Come Forward"… Gangnam Police Station Launches Full Staff Investigation After New Scandal
- "Student ID Rentals Reach 500,000 Won... Black Market and Line-holding Services Surge"
A film formed during charge-discharge processes (redox reactions) between lithium ions and electrolyte. It has very low electronic conductivity but high lithium ion conductivity, behaving like a solid electrolyte. When thin, it acts like a separator, improving battery efficiency; however, if it thickens, it increases internal battery impedance, reduces lithium ion quantity, and causes battery degradation.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![[Image source=Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2021022414102792638_1614143428.jpg)









![[Breaking] Lee Jae-yong: "All Samsung members are united as one... We must pool our wisdom and move in a single direction"](https://cwcontent.asiae.co.kr/asiaresize/307/2026010108513575608_1767225095.png)