Pfizer Vaccine Final Approval 'Imminent'...Medical Community Urges Vaccination for Patients with Underlying Conditions (Comprehensive)
Pfizer Vaccine Final Approval Decision by MFDS on the 5th
AZ Vaccine Death Cases Total 6
Medical Community Voices "Even Patients with Underlying Conditions Should Get Vaccinated"
[Asia Economy Reporters Seo So-jeong and Kim Ji-hee] The final approval of Pfizer's COVID-19 vaccine, 'Comirnaty,' in South Korea will be decided on the 5th. If approved by the final inspection committee of the Ministry of Food and Drug Safety (MFDS) on the morning of that day, it will become the second officially approved COVID-19 vaccine in the country, following AstraZeneca, which was approved on the 10th of last month.
The MFDS will hold the final inspection committee meeting at 10 a.m. in Osong, Chungbuk, to decide on the final approval of the Pfizer vaccine and will announce the results at 2 p.m. The final inspection committee is the last stage of the triple verification system for COVID-19 vaccines and treatments operated by the MFDS. Since both the advisory committee and the central pharmaceutical review committee have already recommended approval for this vaccine, it is expected to pass this final hurdle smoothly. A key point of interest in this approval is whether vaccination will be extended to adolescents aged 16 to 17.
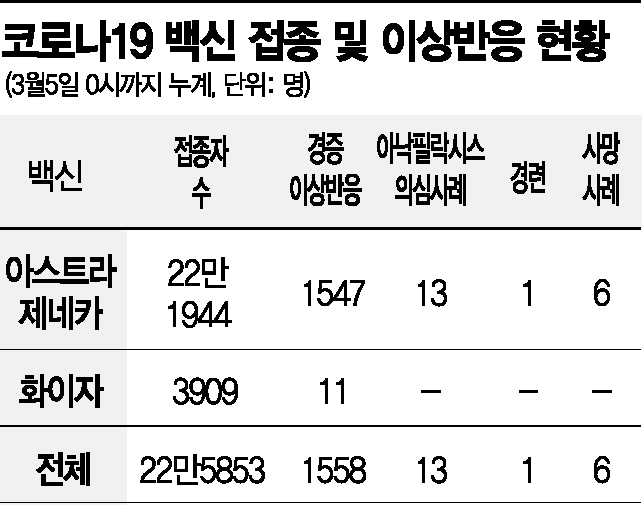
However, even if the vaccine receives approval for use in those aged 16 and older, including adolescents under 18 in the COVID-19 vaccination target group requires a decision from the Korea Disease Control and Prevention Agency's (KDCA) Vaccination Expert Committee. Although the Pfizer vaccine has not yet received formal approval, it is already being administered to healthcare workers treating COVID-19 patients. This supply was imported through the international vaccine procurement project 'COVAX Facility' under a special import approval, and from the 27th of last month until midnight on the 5th, a total of 3,909 people have received the Pfizer vaccine. So far, 11 adverse reaction reports have been confirmed, all of which were mild cases.
According to the COVID-19 Vaccination Response Promotion Team, as of midnight on the 5th, 67,153 people have received additional COVID-19 vaccine doses, bringing the total number of people who have completed the first dose to 225,853. Among them, 221,944 received the AstraZeneca vaccine, and 3,909 received the Pfizer vaccine. As the number of vaccinated individuals increases, 860 additional suspected adverse reaction reports have been filed, totaling 1,578 cases. Of these, 1,558 were mild cases such as headaches, fever, and nausea, and 13 were suspected anaphylaxis cases.
Notably, four additional deaths following vaccination were reported the previous day. Since vaccinations began on the 26th of last month, a total of six deaths have been reported over one week, all among AstraZeneca vaccine recipients. Medical societies including the Korean Diabetes Association, the Korean Academy of Tuberculosis and Respiratory Diseases, and the Korean College of Rheumatology issued a statement recommending vaccination for patients with underlying conditions, emphasizing that although anxiety is spreading due to reported deaths with no confirmed causal relationship to the vaccine, causality between the vaccine and deaths cannot be established.
Furthermore, the government has confirmed the import of 1.05 million doses of the AstraZeneca vaccine through COVAX by May. Prime Minister Chung Sye-kyun stated at the Central Disaster and Safety Countermeasure Headquarters meeting, "The supply schedule for AstraZeneca vaccines to be imported in the first half of the year through the COVAX Facility has been finalized," adding, "Approximately 350,000 doses will be imported this month, and 700,000 doses in April and May, totaling 1.05 million doses." He also instructed, "The KDCA should reschedule to enable as many citizens as possible to be vaccinated in the first half of the year." Regarding the implementation timing of the social distancing system reform plan to be announced at the public hearing that day, he said, "It is difficult to set the application date in advance."
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
Meanwhile, Asan Medical Center in Seoul began its own vaccination program on the morning of the 5th at 8 a.m. for doctors, nurses, and health staff at the COVID-19 staff vaccination room on the 6th floor of the hospital's East Building. COVID-19 vaccinations are scheduled to be administered to about 800 people daily over approximately two weeks from that day until the 19th. The total number of staff targeted for vaccination is about 7,700.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![[Image source=Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2021030115270199450_1614580020.jpg)