"Report on Countermeasures Within 6 Months of Adverse Events in Advanced Biopharmaceuticals"
[Asia Economy Reporter Cho Hyun-ui] In the future, bio companies must report an investigation plan within 15 days and submit a response plan within 6 months if a serious adverse event occurs after the use of their advanced biopharmaceuticals such as gene therapy products.
The Ministry of Food and Drug Safety (MFDS) announced on the 7th, "If a serious adverse event occurs after the use of advanced biopharmaceuticals, thorough investigations and prompt measures such as suspension of use will be taken."
This follows the enactment and full implementation of subordinate regulations under the Advanced Regenerative Bio Act to strengthen the quality and safety management of biopharmaceuticals and support rapid product commercialization.
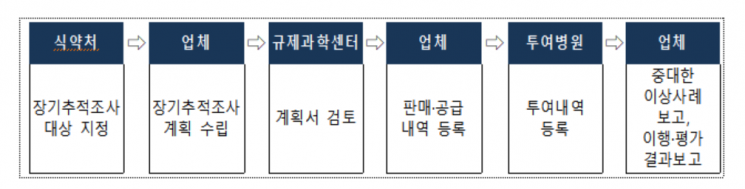
Advanced biopharmaceuticals designated for long-term follow-up investigations require companies to submit a long-term follow-up investigation plan to the MFDS before selling the product.
Physicians administering advanced biopharmaceuticals subject to long-term follow-up must register patient personal information with the Regulatory Science Center for Advanced Biopharmaceuticals after obtaining patient consent, and companies must report the progress of long-term follow-up investigations to the MFDS.
If a serious adverse event occurs, companies must report an investigation plan to the MFDS within 15 days and establish and report the cause, causal relationship with the drug, and countermeasures within 6 months.
The MFDS has established a new industry category specializing in handling human cells used as raw materials for advanced biopharmaceuticals and set detailed standards for the approval process and ensuring the quality and safety of human cells.
To operate cell processing facilities and human cell management businesses, applicants must have facilities, personnel, and equipment and apply for approval. After document review and on-site inspection, approval is granted if deemed appropriate.
Human cell management operators must conduct regular inspections of facilities and equipment and quality tests of human cells, and immediately report to the MFDS if hazardous human cells are discovered.
Currently marketed cell therapy products will be re-verified for safety and quality standards before product approval.
The MFDS will also designate advanced biopharmaceuticals requiring rapid development as "fast-track targets" and support product commercialization through customized and priority reviews from the early development stage.
Fast-track targets include advanced biopharmaceuticals for serious diseases without alternative treatments, rare diseases under the Rare Disease Management Act, and those aimed at preventing or treating pandemic infectious diseases. If designated products present data confirming safety and efficacy from early clinical trial results or during clinical trials, approval will be granted on the condition of submitting subsequent clinical trial data.
Additionally, the MFDS has designated the "Korea Institute of Drug Safety Management" as an institution to provide comprehensive information and technology support for biopharmaceuticals and will establish the "Vaccine Safety Technology Support Center" to support vaccine development.
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
Director Lee Ui-kyung stated, "We will operate a customized approval policy to support the commercialization of biopharmaceuticals with ensured safety and quality," adding, "With the implementation of the Advanced Regenerative Bio Act, we expect to strengthen the quality and safety management of advanced biopharmaceuticals, enhance product competitiveness, and provide new treatment opportunities for patients with rare and intractable diseases."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.