Immune Regulatory Gene That Aggravates Pulmonary Fibrosis Identified
National Institute of Health Confirms Inflammation-Suppressing Role of ATF3 Gene
New Potential Target Suggested for Treating Intractable Pulmonary Diseases
The mechanism behind the progression of 'idiopathic pulmonary fibrosis,' a condition in which the lungs become abnormally stiff for unknown reasons, has been elucidated by a Korean research team.
The National Institute of Health under the Korea Disease Control and Prevention Agency announced on April 28 that it has identified a new function of the 'ATF3 (Activating Transcription Factor 3)' gene, which regulates abnormal immune responses in the progression of idiopathic pulmonary fibrosis.
ATF3 is an activating transcription factor that is triggered when cells experience inflammation or various stress stimuli, and it regulates processes such as metabolism and immune responses in the body.
Idiopathic pulmonary fibrosis is a rare and incurable lung disease of unknown cause, characterized by the gradual hardening of lung tissue and a decline in respiratory function. As the disease progresses, shortness of breath makes daily life difficult, and the prognosis is extremely poor, with death usually occurring within a few years after diagnosis.
Currently, two therapeutic drugs—'pirfenidone' and 'nintedanib'—have been developed and are in use, but they cannot cure the disease and only slow its progression. In response, the National Institute of Health has been conducting research to identify key factors and mechanisms that regulate fibrosis progression.
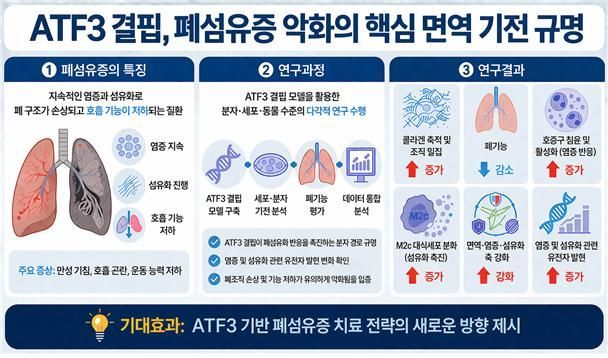
The researchers aimed to determine how ATF3, a transcription factor known to be activated early in response to inflammation or stress, regulates immune mechanisms and pulmonary fibrosis in the lung. To do this, they induced pulmonary fibrosis in an animal model deficient in ATF3.
The results showed that when ATF3 was deficient, lung function deteriorated significantly. In the ATF3-deficient group, lung volume decreased by 20-25% compared to the control group (which showed a decrease of about 15-20%), lung elasticity increased, and lung compliance decreased—demonstrating a pronounced stiffening of the lungs. These findings indicate that ATF3 deficiency can accelerate the progression of pulmonary fibrosis and worsen pulmonary function.
ATF3 deficiency was also found to greatly increase inflammatory responses in lung tissue. Neutrophils, which are responsible for the initial inflammatory response, increased more than 10-fold, and M2c phenotype macrophages—which promote fibrosis—increased 6.5-fold. This resulted in an abnormal composition of immune cells. Additionally, the expression of fibrosis-related genes increased, confirming that both inflammation and tissue damage were exacerbated simultaneously.
Transcriptome analysis revealed that in the ATF3-deficient group, the expression of inflammation- and fibrosis-related genes increased by more than 1.5 times, and immune/inflammation-related pathways were activated. This supports the conclusion that ATF3 plays a critical role in regulating immune responses and suppressing the progression of pulmonary fibrosis.
The research team stated, "We have identified a new molecular mechanism that simultaneously regulates the inflammatory response of immune cells and tissue fibrosis during the progression of pulmonary fibrosis," adding, "It is significant that we confirmed the ATF3 gene, which responds to early stress, plays a key role in suppressing excessive activation of inflammatory responses and mitigating the progression of pulmonary fibrosis."
Kim Wonho, Director of the Department of Chronic Disease Convergence Research at the National Institute of Health, said, "Because pulmonary fibrosis is a chronic lung disease that is difficult to treat, new therapeutic strategies are extremely important," adding, "We will continue to investigate the mechanisms behind chronic respiratory diseases and pursue research that can be applied to actual patient treatment strategies."
Hot Picks Today
 "Forged IDs Easily Bypass Convenience Stores, B...
"Forged IDs Easily Bypass Convenience Stores, B...
- "Pay for the Postpartum Care Center with My Card"... Grandparents Open Their Wal...
- "Skipping Diamond Rings... Gen Z in the US Opts for Finger Tattoos"
- Labor Day and Constitution Day Designated as Public Holidays... Approved by Cabi...
- Back After 3 Years and 5 Months with a 'Shark Nose Surgery'... Hyundai Unveils T...
This research was published in February in 'Clinical Science,' a leading international journal in the field of allergy and immunology.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.