Celltrion Launches Aptosma as First Mover in Japan
Expansion of Autoimmune Disease Portfolio
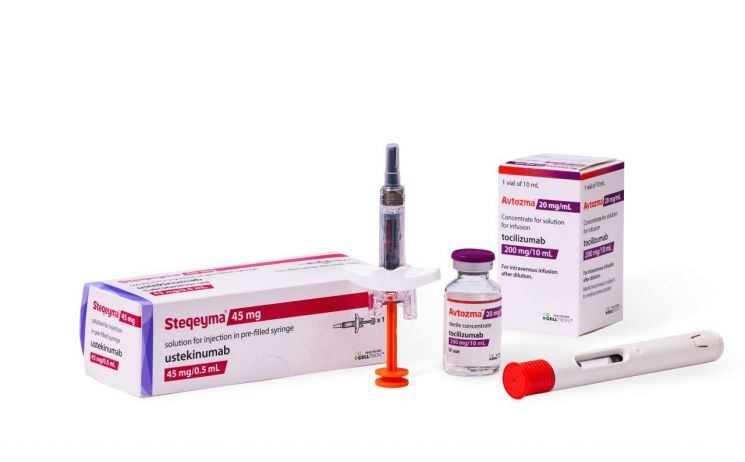
Celltrion announced on April 28 that it has launched 'Aptosma' (ingredient: tocilizumab), a treatment for autoimmune diseases, in Japan as a 'First Mover' (the first product to be released in the category).
Aptosma was launched approximately seven months after obtaining product approval from the Japanese Ministry of Health, Labour and Welfare (MHLW) in September of last year. At the time of approval, Celltrion received authorization for major indications held by the original product, including rheumatoid arthritis (RA), juvenile idiopathic arthritis (JIA), Castleman disease (CD), and cytokine release syndrome (CRS).
Celltrion's Japanese subsidiary plans to directly market (direct sales) Aptosma and aims for an early market settlement by leveraging its accumulated sales expertise and supply capabilities from existing products.
As part of this strategy, prior to Aptosma’s launch, Celltrion's Japanese subsidiary provided academic information about the product to medical professionals at the '70th Annual Meeting of the Japan College of Rheumatology,' and introduced the features of the product based on clinical data to increase interest among the medical community.
Celltrion continues to achieve stable sales performance in Japan’s autoimmune disease market. Its flagship autoimmune disease treatments, 'Remsima' (ingredient: infliximab) and 'Yuflyma' (ingredient: adalimumab), maintain leading positions in biosimilar prescriptions in Japan with market shares of 44% and 19%, respectively, according to IQVIA and other market data. In addition, 'Stekima' (ingredient: ustekinumab), which was launched in August of last year, is also showing steady growth. Celltrion plans to leverage the strength of its expanded four-product autoimmune disease portfolio to conduct marketing activities targeting local medical institutions.
Celltrion also plans to further expand its autoimmune disease portfolio in Japan in the future. 'Omriclo' (ingredient: omalizumab), which received product approval in Japan this March, is being prepared for local launch in the second half of the year. The company also plans to introduce 'Remsima SC,' the world’s only subcutaneous formulation of infliximab, to the Japanese market.
Autoimmune diseases typically require long-term treatment, and a variety of treatment options are often used depending on the patient’s condition. Having a diverse range of therapies enables more tailored responses to individual treatment situations and enhances expertise in the disease area. By expanding its autoimmune disease product portfolio, Celltrion aims to strengthen its competitiveness and achieve greater results in the Japanese market.
A Celltrion representative stated, “With the launch of Aptosma, our autoimmune disease portfolio in Japan now comprises four products, and we anticipate even greater synergy and enhanced business competitiveness among our offerings. By leveraging the experience and sales infrastructure built through existing products, we will provide a stable supply of Aptosma and further strengthen our presence in the Japanese market.”
Hot Picks Today
 "Pay for the Postpartum Care Center with My Car...
"Pay for the Postpartum Care Center with My Car...
- "Forged IDs Easily Bypass Convenience Stores, Bars, and Motels... Delinquency En...
- Unable to Use Photoshop... How a Nordic Aesthetic Cosmetics Commercial Was Made ...
- General Hong Beom-do's Birth Information Standardized as "August 27, 1868, Pyong...
- Back After 3 Years and 5 Months with a 'Shark Nose Surgery'... Hyundai Unveils T...
Meanwhile, Aptosma is an interleukin (IL) inhibitor that suppresses the action of interleukin-6, which is involved in causing inflammation in the body, thereby reducing inflammation. Last year, it achieved global sales of approximately 2.47 billion Swiss francs (about KRW 3.7 trillion). According to IQVIA, the Japanese tocilizumab market was estimated to be worth about USD 337.6 million (approximately KRW 470 billion) as of last year.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.