Biopharmaceutical Clinical Trial Approvals Surge 24% in One Year
Anticancer Drug Clinical Trials Up 10% Year-on-Year, Says Ministry of Food and Drug Safety
Expansion of Multinational Trials Led by Overseas Pharmaceutical Companies
Improved Access to New Drugs for Domestic Patients
As the enthusiasm for new drug development in the domestic pharmaceutical and bio industry intensifies, the number of clinical trial approvals last year saw a slight increase compared to the previous year. Growth was particularly notable in the biopharmaceutical and anticancer drug sectors, and multinational clinical trials targeting global new drugs were also actively conducted.
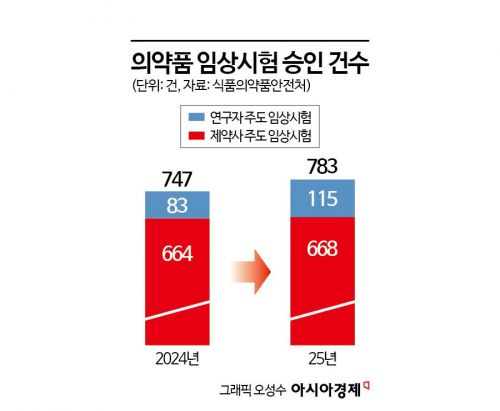
According to an analysis of the pharmaceutical clinical trial approval status released by the Ministry of Food and Drug Safety on March 6, the total number of approvals last year was 783, representing a 4.8% increase from 747 in 2024.
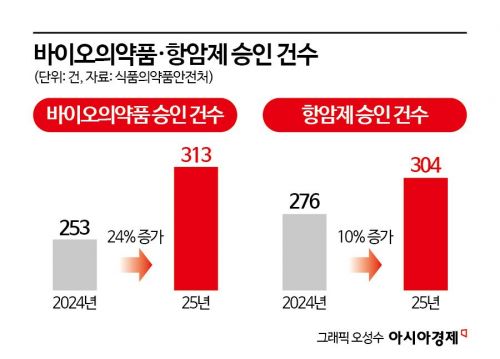
Clinical trials for biopharmaceuticals, including gene-recombinant drugs, surged by about 24%, rising from 253 cases in 2024 to 313 last year. This reflects the trend of expanding market share for the biopharmaceutical sector in the global pharmaceutical market, which has clearly manifested in clinical trial activity in Korea.
Approvals for anticancer drug clinical trials totaled 304 cases, a 10% increase from the previous year. Notably, targeted anticancer drugs accounted for an overwhelming 68% (207 cases) of all anticancer drug clinical trials. This outcome is attributed to the global trend of expanding drug candidates for various types of cancer, including those with specific molecular alterations, combined with Korea's outstanding capabilities in conducting clinical trials.
By trial sponsor, pharmaceutical company-led trials accounted for 668 cases, making up 85.3% of the total. However, investigator-initiated trials conducted by university hospitals and similar institutions also rose sharply to 115 cases, a 38.6% increase from the previous year.
Multinational clinical trials, regarded as a key indicator of global new drug development, recorded 425 cases, up approximately 14% from the previous year. As clinical trials for drugs developed by overseas pharmaceutical companies increased to 409 cases, opportunities are expected to expand for Korean patients to access the latest promising new drugs from abroad more quickly.
In contrast, clinical trials for drugs developed by domestic pharmaceutical companies decreased by about 15%, totaling 259 cases. However, trials utilizing next-generation technologies such as bispecific antibodies, antibody-drug conjugates (ADCs), and live biotherapeutics remain active.
Hot Picks Today
 Frozen Meals, Two Hours of Sleep... "I Wish I C...
Frozen Meals, Two Hours of Sleep... "I Wish I C...
- The Quoted 800,000 Won, the Bill Was 5 Million... Bereaved Families of 'No-Funer...
- "You Can Only Have This in Korea": Which National Museum Cafe Menu Is Captivatin...
- Tragedy in Luxury Apartment: Woman in Her 20s Found Dead, Suspect Identified
- "Never Hike Alone as a Woman" "Even Two Are at Risk"... Growing Fear of Crime on...
The government plans to continue developing improvement measures to enhance consistency and predictability in reviews through the currently operating 'Regulatory Innovation Meeting on Clinical Trial Approvals.' An official from the Ministry of Food and Drug Safety stated, "We will actively pursue clinical trial approval policies that improve patient access to new drugs and support our companies in developing globally competitive new drugs."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.