Lithium-Air Battery "Rapid Charging" Achieved... 2D Catalyst Enhances Both Lifespan and Power Output [Reading Science]
Activating the Entire Inactive Basal Plane Through Atomic Defect Engineering
Stable High-Speed Charge-Discharge for 550 Cycles
A catalytic technology that can simultaneously solve the issues of rapid charge-discharge and short lifespan in lithium-air batteries—often referred to as "dream batteries" for their potential to dramatically extend electric vehicle driving range—has been developed by a Korean research team. The key lies in designing atomic-level defects to convert the entire basal plane of a two-dimensional material, which previously barely participated in reactions, into a catalytic active site.
The joint research team led by Senior Researcher Sohee Jung of the Extreme Properties and Materials Research Center at the Korea Institute of Science and Technology (KIST) and Principal Researcher Kwanghee Lee of the Center for Advanced Materials Processing at the Institute for Advanced Engineering (IAE) has developed a catalytic technology that maximizes the surface activity of the two-dimensional nanomaterial tungsten diselenide (WSe₂). As a result, they have simultaneously improved both the performance and durability of lithium-air batteries.
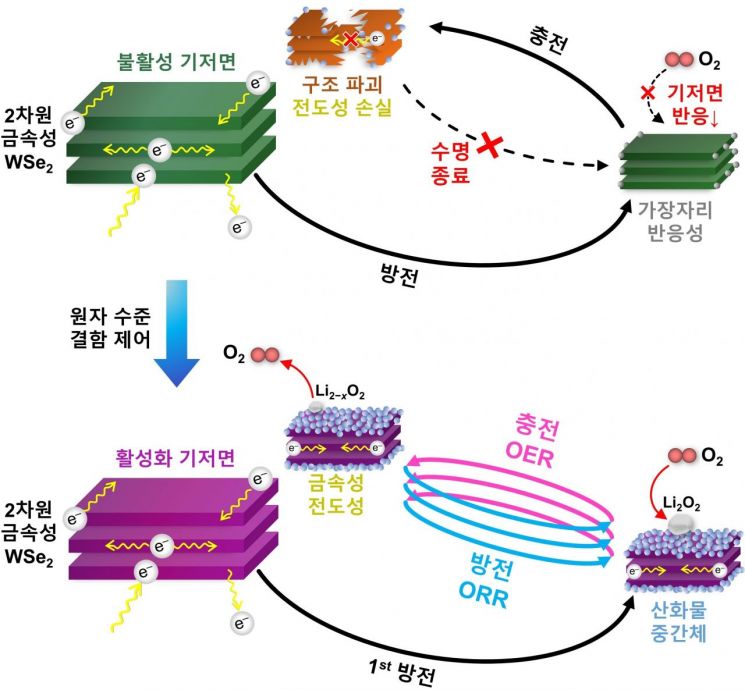
Activation of Basal Plane and Catalytic Reaction Mechanism of Two-Dimensional Metallic WSe2. Two-dimensional metallic WSe2 exhibits catalytic reactions limited only to the edge sites, restricting its performance. However, with atomic-level defect engineering through platinum substitution and selenium vacancies, the entire basal plane becomes activated. At this time, the high reactivity of the oxide intermediates combined with the electrical conductivity of the metallic material maintains a stable charge-discharge cycle. Provided by the research team
View original imageLithium-air batteries are considered next-generation batteries because, in theory, they can achieve energy densities more than 10 times higher than conventional lithium-ion batteries. However, a key challenge in commercialization has been that catalytic active sites facilitating the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) during charge and discharge are limited, resulting in slow reaction rates and short lifespans.
Atomic Defect Engineering Revives the Inactive Basal Plane
The research team intentionally introduced atomic-level defects—specifically, selenium (Se) vacancies—by substituting platinum (Pt) atoms into the layered structure of metallic two-dimensional WSe₂. These defects serve as reaction sites that strongly adsorb and activate oxygen molecules, playing a key role in lowering the energy barriers for the ORR and OER.
What sets this research apart is the comprehensive conversion of the "basal plane," which makes up most of the two-dimensional material’s surface and was previously chemically inactive, into a catalytic active region. The internal metallic structure maintains electrical conductivity, while surface oxide intermediates drive catalytic reactions, resulting in a "synergistic structure," according to the research team.
Furthermore, the team developed a process in which platinum substitution and defect formation can occur through a simple stirring method at room temperature, reducing both process complexity and material damage compared to existing basal plane activation techniques.
Stable for 550 Cycles Even at 1C Rapid Charging... Outperforms Commercial Catalysts
Lithium-air batteries utilizing this catalyst achieved stable lifespans exceeding 550 cycles under high-speed charge-discharge conditions of 1C (1000 mA/g). The batteries also demonstrated superior stability and durability compared to commercial catalysts such as Pt/C and RuO₂ across a range of charge-discharge rates from 0.1C to 3C. This outcome is seen as evidence of the potential to realize next-generation high-power energy systems with minimal performance degradation under rapid charging conditions.
Senior Researcher Sohee Jung stated, "It is significant that we have proposed a strategy to utilize the basal plane, which could not be exploited until now, by controlling it at the atomic level while preserving the structural advantages of two-dimensional materials." Principal Researcher Kwanghee Lee added, "By overcoming the challenge of rapid charge-discharge performance in lithium-air batteries, we can accelerate the commercialization of high-power mobility energy systems."
The researchers believe that this technology can be applied not only to lithium-air batteries but also to a variety of energy conversion and storage fields requiring high-performance catalysts, such as water electrolysis and fuel cells. Notably, participation from Lawrence Livermore National Laboratory (LLNL) in the United States has enhanced the reliability and global collaboration of the research.
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
This research was published in the latest issue (January 19) of the international journal "Materials Science and Engineering: R: Reports." The study was supported by the Ministry of Science and ICT and conducted through major KIST projects, individual basic research programs, the Future Pioneering Convergence Science & Technology Development Program, and the Global TOP Strategy Research Group project.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







