Novo Nordisk's Next-Generation Obesity Drug Surpasses GLP-1 Limits in Patients With Diabetes
4 Percentage Points Greater Weight Loss Than Semaglutide in Phase 3 Trial
Demonstrated Sustained Glycemic Control and Reduced Weight-Loss Plateau
Stronger Competitiveness in Diabetes Market, Where Reimbursement Barriers Are Lower Than for Obesity Treatments
Novo Nordisk's next-generation obesity and diabetes drug has demonstrated greater weight-loss effects and stable blood sugar control compared with existing glucagon-like peptide-1 (GLP-1) agonist therapies in a phase 3 clinical trial in patients with type 2 diabetes. Attention is now focused on whether the company can overcome its weak performance in obesity trials and rebuild competitiveness in the diabetes treatment market.
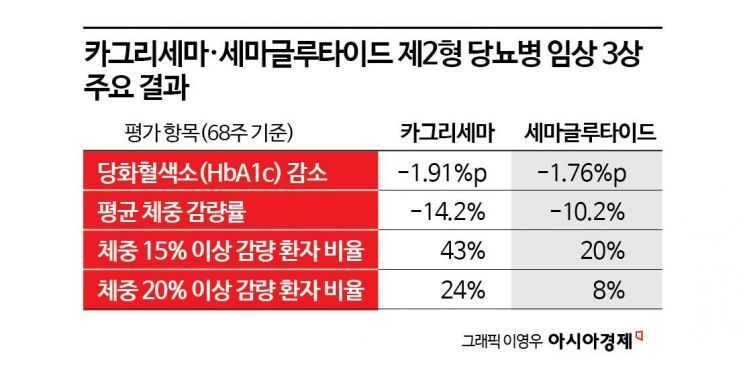
According to the global phase 3 CagriSema trial results released by Novo Nordisk on the 4th, the study was conducted over 68 weeks in more than 2,700 patients with type 2 diabetes. The average weight-loss rate in the CagriSema arm was 14.2%, exceeding the 10.2% seen with semaglutide monotherapy by 4 percentage points. The blood sugar marker HbA1c was also similar to, or better than, that of the comparator group.
Weight loss in patients with diabetes is structurally more difficult than in patients with obesity. This is because they have higher insulin resistance, and as blood sugar improves, the loss of glucose in the urine decreases, which in turn increases energy conservation. Even Mounjaro, currently the obesity treatment with the greatest weight-loss effect, showed more than 20% weight reduction in obesity trials, but only slightly above 10% in diabetes trials.
CagriSema secures blood sugar stability by simultaneously stimulating GLP-1 and the amylin hormone. While the GLP-1 receptor agonist directly lowers blood sugar by promoting insulin secretion, amylin slows gastric emptying and blunts the sharp rise in postprandial glucose, creating a dual mechanism. This "cooperative action" not only controls blood sugar, but also amplifies powerful satiety signals through different pathways in the brain. As a result, it can increase the degree of weight loss in patients with diabetes while maintaining stable metabolic control.
In a phase 3 trial in patients with obesity released in 2024, CagriSema achieved an average weight-loss rate of 22.7%, falling short of the roughly 25% reduction the market had anticipated. Because this outcome failed to meet investors' expectations that it would outperform the competing drug Mounjaro, Novo Nordisk's share price plunged immediately after the trial results were released. Given that weight loss is structurally more difficult in patients with diabetes than in those with obesity, analysts say the meaningful effect confirmed in this trial supports the need to reassess CagriSema's clinical and commercial competitiveness.
From the standpoint of market uptake, the diabetes treatment market is considered to offer a more advantageous position than the obesity treatment market. While the obesity treatment market has greater growth potential, in most countries obesity drugs still face high reimbursement hurdles, leaving patients with a heavy out-of-pocket burden. In contrast, diabetes, as a chronic disease, already has a well-established reimbursement framework, so once clinical differentiation is demonstrated, a broad and stable prescribing base can be secured in a relatively short period of time.
Hot Picks Today
!["What Should I Eat? Cooking at Home Is a Hassle... 10,000~20,000 Won 'Hotel-Level Catering' Spreads in Ultra-Luxury Apartments [Luxury World]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026042610544940447_1777168490.png) "What Should I Eat? Cooking at Home Is a Hassle...
"What Should I Eat? Cooking at Home Is a Hassle...
- Even with KOSPI at 6,500..."Selling Samsung and SK hynix for Cash," Individuals ...
- "Up to 600,000 Won Per Person, Finally Available"... Be Careful: Filling Up at Y...
- "Survived Thanks to Korean Choco Pie"...How a Vietnamese University Student Endu...
- No Work, No Inheritance for the Eldest... 30 Billion KRW in Shares Gifted to Sec...
Whether a drug is covered by insurance is cited as a key variable for expansion of the GLP-1 therapy market. According to a paper published last year by U.S. researchers in the international journal JAMA Health Forum, if public insurance were to include GLP-1 obesity treatments in its reimbursement list, drug expenditures would increase by about 65.9 billion dollars (approximately 95 trillion won) over the next 10 years. The analysis concludes that expanded insurance coverage directly translates into increased prescriptions and higher sales.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.