UNIST Develops Eco-Friendly Production Technology for Propylene Oxide, a Key Raw Material for Mattresses and Fabrics
Self-Generating Hydrogen Peroxide Enables Self-Driving System for Propylene Oxide Production
New Catalyst Structure Boosts Productivity Eightfold... Published in Nature Communications
A self-driving system that produces propylene oxide in an eco-friendly manner without electricity or solar energy has been developed.
Propylene oxide is a key raw material used in the production of everyday materials such as polyurethane, which is found in sofas and mattresses, and polyester, which is used in fabrics and water bottles.
A research team led by Professors Kwak Jahoon and Jang Jiuk from the Department of Energy and Chemical Engineering at UNIST, along with Professor Cho Sungjun from Chonnam National University, announced on October 23 that they have developed a self-driving system that produces propylene oxide using hydrogen peroxide generated within the system itself.

Research team, (from left) Professor Jahun Kwak, Professor Jiuk Jang, Researcher Kwanghyun Kim, Researcher Sunwoo Hwang, Researcher Taehyun Kim. Provided by UNIST
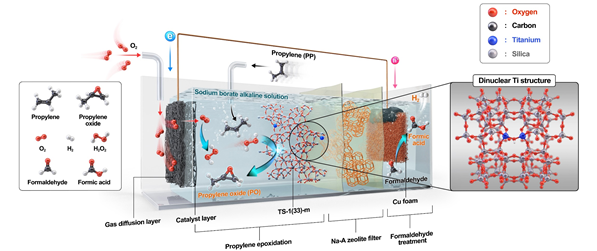
View original imagePropylene oxide is produced by oxidizing propylene, which requires hydrogen peroxide as an oxidizing agent in the production process. Unlike conventional commercial hydrogen peroxide production processes, the newly developed system can generate hydrogen peroxide on-site without emitting pollutants or carbon. The system operates spontaneously without external electricity or solar power by utilizing the electrochemical reactions between oxygen and formaldehyde, taking advantage of the energy difference between these two reactions.
The hydrogen peroxide produced then reacts with separately injected propylene within the system to create propylene oxide. The research team significantly improved productivity by newly designing the structure of the catalyst required for this oxidation reaction.
This addresses the issue of traditional zeolite-based catalysts (TS-1) rapidly losing activity in alkaline environments. While alkaline conditions are essential for efficient hydrogen peroxide production, low catalyst activity in such conditions hampers the subsequent propylene oxidation reaction, ultimately reducing the efficiency of propylene oxide production.
The developed system produced 1,657 micromoles (μmol) of propylene oxide per square centimeter over 24 hours. This is approximately eight times higher productivity than previously developed eco-friendly hydrogen peroxide-based production processes. Additionally, hydrogen (H₂), a clean energy resource, is also produced during the process.
According to an economic analysis, this system can reduce the production cost of propylene oxide by about 8% (to $2.168 per kilogram) compared to currently commercialized production methods.
This is because it does not require complex pre-processing steps or high-temperature, high-pressure equipment, and it operates without external energy, significantly reducing both capital and operating expenses. Since hydrogen peroxide is produced and used directly on-site, transportation and storage costs can also be saved.
Professor Jang Jiuk stated, "As a modular process that can be installed and used wherever needed, this system enables small-scale, site-specific production, which could accelerate the shift from large-scale centralized production to a distributed production system."
Professor Kwak Jahoon commented, "This is a case of gradually overcoming the longstanding technical limitations of traditional zeolite catalysts, and it will serve as an important stepping stone to making the chemical industry much more eco-friendly and sustainable."
A self-driven system that produces propylene oxide without external electricity.
View original imageThis research was supported by the STEAM Research Program of the National Research Foundation of Korea (NRF) under the Ministry of Science and ICT, as well as the Institute for Basic Science (IBS). Equipment support was provided by the UNIST Equipment Education and Support Center and the 6D UNIST-PAL beamline at the Pohang Accelerator Laboratory (PAL).
Hot Picks Today
 Chilling Timing "Did They Know Again?"... $640 ...
Chilling Timing "Did They Know Again?"... $640 ...
- Trump: "Iran in a Crazy Situation... Orders to Fire on Mine-Laying Vessels in th...
- "I Spent Money in a Truly Meaningful Way"... How an SK hynix Employee Donated 10...
- "If You Have This at Home, Remove It Immediately"... 'This Item' Is as Harmful a...
- Foreign Vessels Once Swept Up the Seas Every Season... Now Face Fines of Up to 1...
The research results were published online in the international journal Nature Communications on September 30.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.







