Bio Industry Welcomes Push to Shorten Biosimilar Approval Period
Approval Period Expected to Be Reduced from 406 to 295 Days
MFDS Begins Review System Reform Process
With the government announcing a significant reduction in the approval period for biosimilars, it is expected that biosimilars already recognized in the global market will find it easier to enter the domestic market.
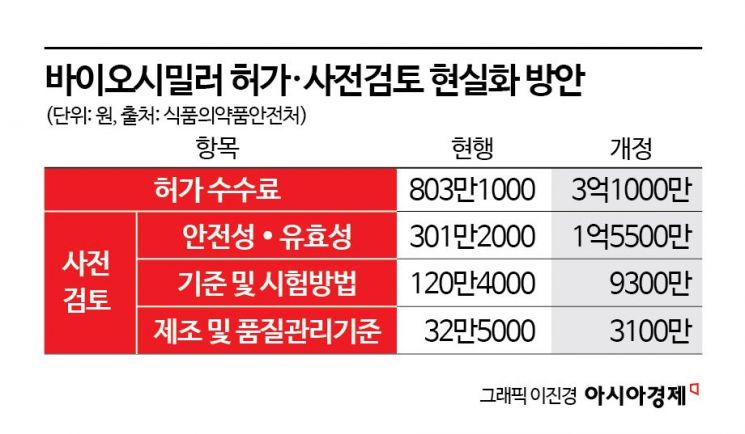
According to the Ministry of Food and Drug Safety (MFDS) on September 15, the statutory processing period for biosimilar approval applications is 115 business days, but in practice, the actual processing time averages 406 days. The MFDS aims to reduce this by more than 100 days, bringing it down to 295 days.
The industry anticipates that the MFDS's measures to shorten the biosimilar approval period will enable faster market entry, leading to a meaningful increase in sales. Korean biosimilar companies such as Celltrion and Samsung Bioepis are already recognized in the global market, recording sales in the trillion-won range in the United States and Europe. This is partly because these markets are larger than the domestic market, but also because governments actively support the distribution of biosimilars, which are 30-40% cheaper than original drugs, through policy initiatives.
An official from a biosimilar development company stated, "It usually takes about a year to obtain product approval in the United States and Europe, but in Korea, it used to take over 400 days, which is longer than the global standard. Although the fees have increased, they remain at an acceptable level, and the increase in sales due to the shortened approval period will be even greater."
Previously, on September 12, the MFDS announced an administrative notice of a partial amendment to the 'Regulations on Fees for Approval of Pharmaceuticals, etc.', which focuses on restructuring the product approval fees for equivalent biological products (biosimilars). An MFDS official explained, "There were issues such as delays in scheduling due to a lack of personnel to conduct manufacturing quality inspections. By adjusting the fees to reflect actual costs, we plan to increase the number of reviewers and reduce the approval period."
This administrative notice is one of the follow-up measures to the 'Bio Innovation Forum,' a meeting between President Lee Jaemyung and the bio industry held on September 5. The goal is to drastically reduce the approval period for biosimilars by applying approval innovation measures, such as fee restructuring for new drugs, to biosimilar approvals as well. According to the revised regulations, the product approval fee for biosimilars has increased from around 8 million won to 310 million won.
The MFDS plans to use the additional funds secured to hire more reviewers, such as doctors and pharmacists, and to operate dedicated review teams. However, to ease the burden on the industry, if a small or medium-sized company applies for approval of a domestically developed biosimilar, 50% of the fee will be waived. If the same applicant submits a similar approval application, the fee for the second and subsequent products will be reduced to 8 million won (based on electronic applications).
In addition to increasing personnel through fee adjustments, a system overhaul is also expected. An MFDS official stated, "We have also initiated procedures to reform the review system, focusing on simplifying processes such as requests for supplementary materials and shortening the supplementation period."
Hot Picks Today
![Home Appliance Woes Mount, Yet 45 Trillion Won in Bonuses? Samsung's Risky Asymmetry [Why&Next]](https://cwcontent.asiae.co.kr/asiaresize/93/2022111609212518207_1668558086.jpg) Home Appliance Woes Mount, Yet 45 Trillion Won ...
Home Appliance Woes Mount, Yet 45 Trillion Won ...
- "Major Crash Is Coming... Buy Even If You Have to Skip a Meal" 'Rich Dad' Shares...
- Concerns Over 'Forever Conflict' Between U.S. and Iran... Brent Crude Surges Pas...
- 500% Energy Efficiency... Samsung Electronics’ 'Energy Magic' to End the Fear o...
- "It Was Fantastic" Jensen Huang's Daughter Seals 'Robot Alliance' with LG throug...
Further efforts by the government to expand the distribution of biosimilars are also anticipated, as the President has personally expressed his commitment. President Lee stated, "The industry has voiced the need to provide incentives to doctors who prescribe affordable and effective medicines, and I believe the Ministry of Health and Welfare can establish such a system. It is a problem that needs to be addressed if doctors continue to prescribe expensive original drugs to patients when the efficacy is the same. This is a form of irrationality," he said.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.