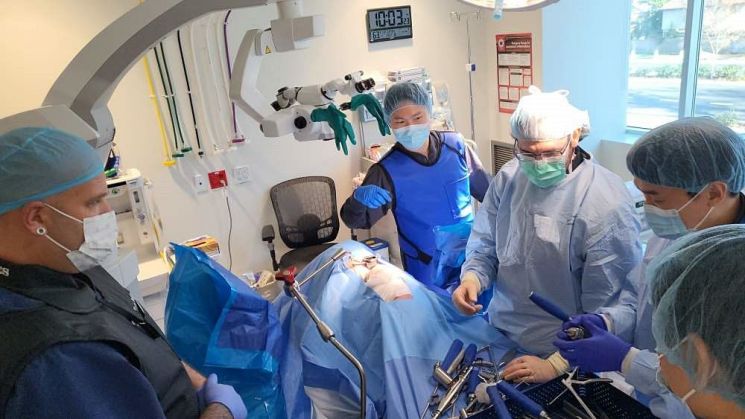
L&K Biomed, Height-Expandable Cage Cadaver Workshop
LNKBioMed announced on the 20th that its U.S. subsidiary, Aegis Spine, held a "Height-Expandable Cage Cadaver Workshop" on the 18th at the Surgical Center in Pocatello, Idaho, USA.
The event was organized at the request of medical professionals interested in height-expandable cage products, including Dr. Karman Pasa and Dr. Tyler Carson. There was significant interest in LNKBioMed's height-expandable product, the "PassLock-TM." Through education on the correct usage, theory, and pre- and post-operative effects of PassLock-TM, the excellence of the product was highlighted. Time was dedicated to providing safe and effective surgeries for patients.
Among the six new products in the "BlueX Series," the "BlueX-LT" and the surgical instrument used together during procedures, the "Retractor," were also introduced. The Retractor is a self-developed surgical instrument used in lateral lumbar interbody fusion (LLIF) and anterior to psoas (ATP) surgeries.
An LNKBioMed representative stated, "We held the cadaver workshop to provide an opportunity for new medical professionals interested in height-expandable cage products to experience the products and for those already using them to demonstrate the new products."
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.