Revealing the Progression and Metastasis Process of Pancreatic Cancer
No Jaeseok Yonsei University Professor Research Team
Helps Develop Epigenetic Therapeutics
Pancreatic cancer is a malignant disease with an average survival period of less than one year. Domestic researchers, through international collaborative research, have uncovered how pancreatic cancer worsens and metastasizes, which is expected to aid in the development of personalized treatment methods.
Yonsei University announced on the 3rd that Professor No Jaeseok's biochemistry research team successfully identified the causative proteins and molecular mechanisms promoting angiogenesis and lung and liver metastasis of pancreatic cancer cells through epigenetic variations in an international collaborative study.
Currently, there are no definitive targeted therapies for pancreatic cancer, and treatment relies on surgical resection or chemotherapy. However, surgical resection is applicable to only a very small number of patients. Chemotherapy treatment also induces drug resistance and cancer recurrence. Therefore, academia and industry worldwide are striving to discover target proteins for developing pancreatic cancer treatments.
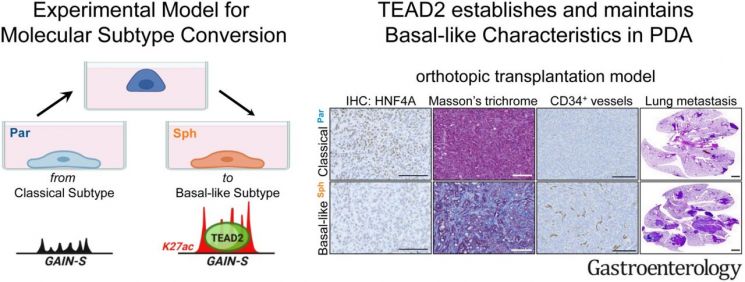
While it is well known that genetic mutations such as KRAS and TP53 are responsible for pancreatic cancer development, the existence of genetic mutations causing malignant progression and metastasis remains controversial. The research team focused on the principle of epigenetic regulation of gene expression called the ‘histone code,’ which is used in somatic cell differentiation, aiming to identify histone codes that promote malignant differentiation of pancreatic cancer cells. To this end, they developed an experimental technique to easily control the malignancy level of pancreatic cancer in vitro and established a research system to track pancreatic cancer occurrence and metastasis in vivo.
Through this, the team sought to examine changes in histone codes and gene expression specifically appearing in malignant pancreatic cancer cells at the single-cell and tissue levels. Unexpectedly, they confirmed that pancreatic cancer cells that had completed malignant differentiation had incorporated histone codes and gene expression programs characteristic of vascular endothelial cells. This showed that these malignant pancreatic cancer cells not only promote angiogenesis of surrounding vascular cells but also form a mesh-like network typical of vascular cells. Animal experiments and analysis of samples from over 100 patients demonstrated that this promotes pancreatic cancer development and metastasis.
This study suggests that lineage changes unique to cancer cells through epigenetic alterations contribute to early cancer occurrence and metastasis, providing clues for the future development of personalized pancreatic cancer therapies. Furthermore, it is expected to help elucidate malignant differentiation processes in various solid cancers such as colorectal, gastric, and liver cancers.
Professor No stated, “Elucidating epigenetic mechanisms involved in metastasis of various solid cancers, including pancreatic cancer, is very important from the perspective of translational basic and clinical research. The significance lies in providing important clues for applying recently spotlighted epigenetic cancer therapies to pancreatic cancer as well.”
Hot Picks Today
 "I Wish I Could Sleep": Frozen Meals, Two Hours...
"I Wish I Could Sleep": Frozen Meals, Two Hours...
- The Quoted 800,000 Won, the Bill Was 5 Million... Bereaved Families of 'No-Funer...
- "You Can Only Have This in Korea": Which National Museum Cafe Menu Is Captivatin...
- Tragedy in Luxury Apartment: Woman in Her 20s Found Dead, Suspect Identified
- "Never Hike Alone as a Woman" "Even Two Are at Risk"... Growing Fear of Crime on...
The research results were published online on the 10th of last month in the international journal in the field of gastrointestinal cancer, Gastroenterology (IF 33.9). (Paper title: A TEAD2-driven endothelial-like program shapes basal-like differentiation and metastasis of pancreatic cancer).
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.