'EV Battery War'... Found a Way to Increase Efficiency and Reduce Process Costs
UNIST Professor Jae-Pil Cho's Team Develops Easy Single Crystal Material Synthesis Technology
Improves Cathode Synthesis Efficiency Using Eutectic Composition Principle... Published in Nature Energy
A method to significantly improve the efficiency of electric vehicle batteries has emerged, attracting global attention. This is because it is evaluated to hold technological value that predicts future dominance in the global market during the 'battery war' era.
Recently, a technology has been developed that can produce complete single-crystal forms of not only nickel-rich cathodes, which are key to increasing the driving range of electric vehicles by more than 30%, but also lithium- and manganese-rich cathode materials at low cost.
The research was conducted jointly by Professor Jae-Pil Cho's team (first author Dr. Moon-Soo Yoon) from the Department of Energy and Chemical Engineering at UNIST (President Yong-Hoon Lee) and Professor Ju Li's team at MIT (Massachusetts Institute of Technology) in the United States.
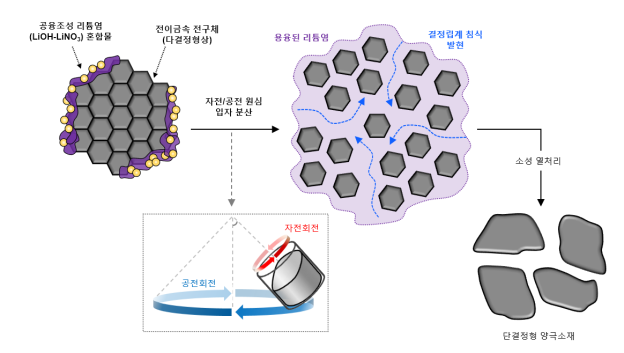
Schematic diagram of LiOH-LiNO3 lithium salts and air/rotation mixing for single-crystal cathode synthesis.
View original imageAccording to the joint research results, the team dissolved lithium hydroxide (LiOH) and lithium nitrate (LiNO3) through a eutectic composition. The dissolved lithium salts and transition metal precursors were mixed using a planetary centrifugal mixer to synthesize a liquefied lithium salt-transition metal nanoparticle composite.
The team developed a core technology that allows heat treatment of this composite below 800 degrees Celsius to produce fully formed single-crystal particles.
Nickel-rich cathode materials, known as high-capacity battery cathode materials, can exhibit high capacity. However, repeated charging and discharging cause microcracks inside the particles, leading to side reactions with the battery electrolyte and a rapid decrease in battery life. Currently commercialized cathode materials are in a 'polycrystalline form' composed of particles clustered at the scale of several hundred nanometers, making it impossible to avoid this phenomenon.
Polycrystalline materials tend to break easily during battery manufacturing and promote unnecessary reactions within the battery. This increases gas generation and affects charge-discharge cycles, reducing battery lifespan.
However, manufacturing cathode materials in a 'single-crystal form' can effectively reduce these problems. The downside is that the processing cost is more than 30% higher compared to polycrystalline materials of the same composition.
The processing cost is important because the price share of cathode materials in an electric vehicle is about 15%, and processing costs account for approximately 2.25% of that. Since metal prices are determined by international market rates, minimizing processing costs is crucial to gaining a price competitiveness advantage.
First, the research team combined lithium nitrate and lithium hydroxide dissolved in a eutectic composition with polycrystalline transition metal precursors in a fixed ratio. They mixed this using a planetary centrifugal mixer at 2000 rpm for 12 minutes.
They demonstrated that the molten powders generated heat upon contact and penetrated the grain boundaries of the polycrystalline particles (manifesting grain boundary erosion), forming a liquefied lithium salt-transition metal nanoparticle composite.
The team developed a technology to heat this composite below 800 degrees Celsius for 10 hours to produce fully crystallized single-crystal particles of several micrometers in size.
This technology can be applied not only to nickel-rich cathodes but also to lithium- and manganese-rich cathode materials. Lithium- and manganese-rich cathodes contain more than 60% manganese (Mn) in high concentration. Additionally, the lithium content is higher than that of transition metals, enabling high capacity of over 250 mAh/g at high voltages above 4.5 V.
As the Mn content increases, the heat treatment temperature required for synthesis rises. Especially when Mn content exceeds 60%, synthesis into single crystals is impossible even after heating above 900 degrees Celsius for more than 12 hours. However, this study revealed that synthesis into single-crystal particles of 1 μm size is possible even with Mn content above 60%.
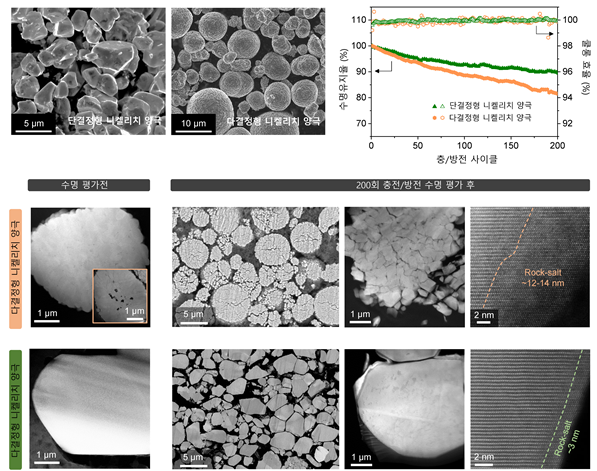
Study on Morphological Changes and Room Temperature Electrochemical Performance Comparison Before and After 200 Cycle Life Evaluation of Single-Crystal NCM811 Nickel-Rich Cathode Material Figure.
View original imageThe research team applied this study to measure battery performance. They evaluated single-crystal cathode materials of the same composition as polycrystalline materials (NCM811) synthesized by conventional processes in lithium metal batteries.
This single-crystal cathode material maintained about 92% of its original capacity even after 200 charge-discharge cycles and showed approximately 12% improved lifespan retention compared to polycrystalline materials of the same composition. It also demonstrated more than 30% improvement in gas generation and resistance increase rates, suggesting a significant role in improving the safety issues that have long plagued electric vehicles.
Dr. Moon-Soo Yoon, first author and postdoctoral researcher at UNIST's Department of Energy Engineering, explained, "Through this research, we revealed that lithium- and manganese-rich cathode materials, which are attracting attention as alternatives to LFP, can be synthesized cheaply in single-crystal form as well as nickel-rich cathode materials."
Professor Jae-Pil Cho of the Department of Energy and Chemical Engineering pointed out, "Currently commercialized nickel-rich single-crystal cathode materials face increased production costs due to multiple heat treatments."
He added, "Applying the newly developed synthesis method to cathode materials is expected to reduce costs by at least 30% compared to existing single-crystal materials when developing mass production processes. However, the current synthesis scale is at the lab level, and it will take at least four more years to achieve mass production."
Hot Picks Today
 "Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
"Buy on Black Monday"... Japan's Nomura Forecasts 590,000 for Samsung, 4 Million for SK hynix
- "Plunged During the War, Now Surging Again"... The Real Reason Behind the 6% One-Day Silver Market Rally [Weekend Money]
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- "We're Now Earning 10 Million Won a Month"... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Are Already Watching Closely..."Target Stock Price 970,000 Won" Now Only the Uptrend Remains [Weekend Money]
This research was published on March 30 (local time) in Nature Energy, a prestigious journal in the energy field.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.








