Seegene Launches World's First Commercial Product with '3 Ct' Technology
[Asia Economy Reporter Lee Chun-hee] Seegene has developed a new product, 'Allplex Inyudojong Virus (HPV) HR Detection,' which commercializes the world's first '3 Ct' technology.
Seegene announced on the 26th that it will showcase the Allplex HPV HR Detection applying 3 Ct technology at the '2022 European Congress of Clinical Microbiology and Infectious Diseases (ECCMID 2022)' held from the 23rd to 26th (local time) in Lisbon, Portugal.
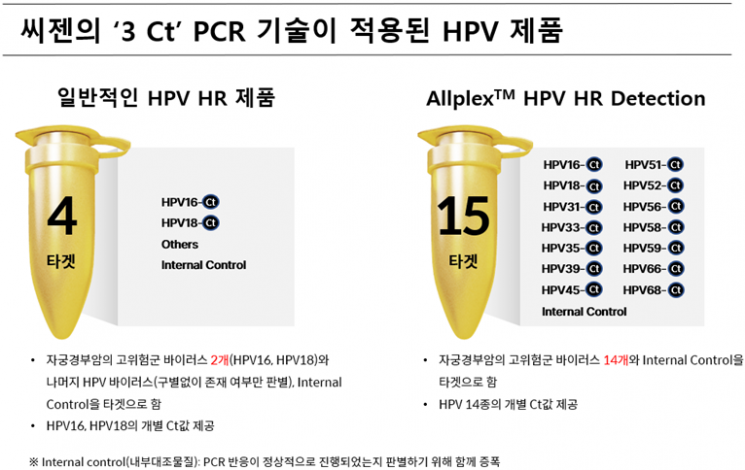
The Ct value refers to the quantitative indicator of the infectious agent (target) in genetic amplification (PCR) testing. Usually, one Ct value is calculated per one target in a single channel. However, Seegene succeeded in implementing three Ct values in one channel while maintaining accuracy by utilizing its molecular diagnostic expertise and patented technology. This allows quantitative information on 15 targets using five channels in one tube.
Seegene plans to apply 3 Ct technology not only to existing products for respiratory diseases (RV), sexually transmitted infections (STI), gastrointestinal infections (GI), and urinary tract infections (UTI) but also to future products. This is expected to upgrade the level of 'syndromic' testing, which simultaneously tests multiple pathogens causing similar symptoms.
It can accurately identify the causative pathogens and whether there is a co-infection, and determine treatment priorities based on the degree of infection, providing great assistance in medical settings. Additionally, the same equipment can perform three times the amount of testing, enabling more efficient large-scale testing. Seegene expects that the spread of 3 Ct technology will significantly improve the service and cost structure of the medical industry.
The new product Allplex HPV HR Detection introduced by Seegene through 3 Ct technology targets 14 high-risk HPV types that can cause cervical cancer. While other companies' products provide individual Ct values only for two types, HPV16 and HPV18, Allplex HPV HR Detection quantitatively confirms the infection level of each of the 14 types through individual Ct values, playing a significant role in the prevention and follow-up management of cervical cancer.
Based on its technological competitiveness, Seegene plans to set an affordable price and apply it to the automated testing equipment 'All In One System (AIOS)' within the first half of the year, aiming to establish the global molecular diagnostics industry's first 'large-scale automated syndromic testing system.' This will provide an environment where anyone can easily receive necessary tests not only in large hospitals or contract testing laboratories (C-Lab) but also in small and medium-sized hospitals, accelerating the 'popularization of molecular diagnostics.'
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
Sebastian Hantz, a professor at the University of Limoges Medical School in France and an HPV expert, explained, "It is essential for patient management to identify which HPV virus types are detected, whether they persist or have disappeared, and to monitor them accordingly." He added, "Syndromic testing can be very useful in clinical situations such as respiratory diseases."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.