NKMAX "Super NK and Keytruda Combination Achieves Complete Remission in Advanced Sarcoma Patients"
[Asia Economy Reporter Lee Gwan-joo] NKMax announced on the 14th that its U.S. subsidiary NKGen Biotech. found that cancer completely disappeared in a terminal sarcoma patient treated under compassionate use approval.
Previously, NKMax revealed in December last year that the tumor in the patient's liver had regressed by about 80%. This patient, diagnosed with desmoplastic small round cell tumor (DSRCT), a type of sarcoma, received treatment combining SuperNK (SNK01) and Merck's Keytruda (Pembrolizumab) under compassionate use approval from the U.S. Food and Drug Administration (FDA).
At the time of the sarcoma diagnosis in 2017, the cancer had metastasized throughout the body, including the liver, lungs, abdomen, and bladder. For about one and a half years before SuperNK administration, chemotherapy and immuno-oncology drugs were administered, but due to toxic reactions and continued tumor growth, treatment was discontinued. Notably, this patient was PD-L1 negative.
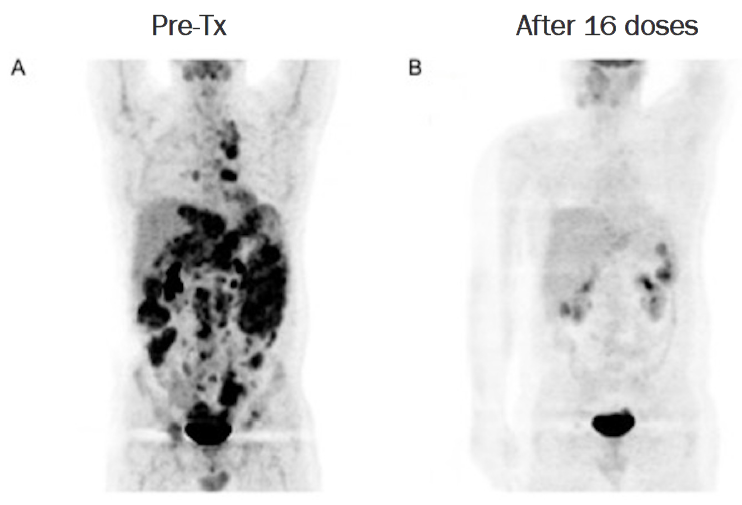
Comparison photos of tumor disappearance before (A) and after (B) combined administration of Super NK (SNK01).
View original imageSubsequently, the patient was registered as a compassionate use case and received a total of 30 combined doses of SuperNK and Keytruda over 28 months. According to PET-CT scans, no residual tumors were observed, indicating the patient reached a complete remission (CR) state, according to the company.
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
Professor Kim Hye-ryun of Yonsei Cancer Center's Department of Oncology stated, "It is a rare case to see a treatment response in a patient who is PD-L1 negative with almost no PD-L1 expression. It is known that only 25-30% of all cancer patients are PD-L1 positive, while 70-75% are PD-L1 negative. If SuperNK can convert these PD-L1 negative patients into positive ones, it could lead to a significant change in the cancer treatment market."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.








