Authorities: "Omicron Will Soon Dominate... Exploring New Response Measures"
Oral Treatment "Mechanism Suggests Efficacy Maintained Against Omicron"
[Asia Economy Reporter Chunhee Lee] Health authorities are considering related response measures as they anticipate that the Omicron COVID-19 variant will soon become the dominant strain domestically.
On the afternoon of the 6th, Park Young-jun, head of the epidemiological investigation team at the Central Disease Control Headquarters (CDCH), stated at the regular COVID-19 briefing, "In January, and likely by February, more than half of all variant analyses will be of the Omicron variant," indicating a high possibility that Omicron will soon become the dominant strain.
The Omicron variant is known to be 3 to 4 times more transmissible than the existing Delta variant. While its virulence is lower, leading to expectations of reduced severity and fatality rates, concerns are greater that the absolute number of severe cases and deaths may increase due to its rapid transmission. Kim Ki-nam, head of the COVID-19 Vaccination Response Team’s Vaccination Management Division, also warned, "If the highly transmissible Omicron variant becomes dominant, confirmed cases could surge rapidly in a short period," adding, "This could lead to increased infections among high-risk groups and raise the overall risk of severe illness in society."
Regarding this, Ko Jae-young, spokesperson for the Korea Disease Control and Prevention Agency (KDCA), said, "It is inevitable that Omicron will become the dominant strain with the increase in confirmed cases, and it is difficult to continue strengthening social distancing measures. Therefore, we are redesigning the overall quarantine and medical response system to actively respond to the Omicron variant," and announced that new response measures will be devised and announced as soon as possible.
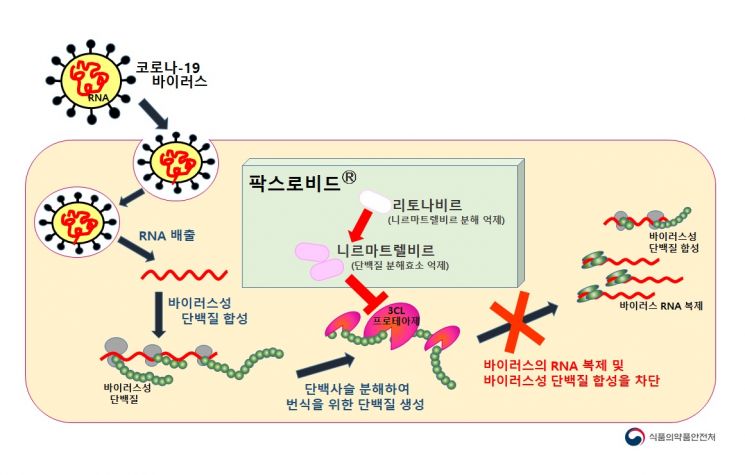
Mechanism of action of Pfizer's oral COVID-19 treatment 'Paxlovid' (Photo by Ministry of Food and Drug Safety)
View original imageHealth authorities believe that the introduction of oral antiviral treatments will help to some extent in overcoming the rapid spread of the Omicron variant. Spokesperson Ko explained, "There has been no specific announcement yet on the efficacy of Paxlovid against the Omicron variant, and research is ongoing," but added, "Based on current information, experts believe that the mechanism of action suggests that the Omicron variant will not affect the protease inhibitor, so its efficacy is expected to be maintained."
Unlike existing vaccines or antibody treatments that inhibit viral replication by binding to the spike protein region of the COVID-19 virus, Pfizer’s Paxlovid blocks the protease enzyme (3CL protease), preventing the production of proteins necessary for viral replication, thereby inhibiting virus proliferation. Since viral mutations typically concentrate on the spike protein, it is expected that efficacy will continue against most variants.
The government currently has purchase contracts for a total of 1,004,000 courses of oral antiviral treatments, including 762,000 courses of Paxlovid and 242,000 courses of Merck & Company (MSD)’s Molnupiravir. Among these, Paxlovid has received emergency use authorization from the domestic Ministry of Food and Drug Safety, while Molnupiravir is still under review.
Hot Picks Today
 "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
"Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- "Not Everyone Can Afford This: Inside the World of the True Top 0.1% [Luxury World]"
- While All Eyes Were on Samsung and Hynix, This Company Surged 50% to New Highs in Four Days [Weekend Money]
- Police to Drastically Advance Protection for Ruling and Opposition Party Leaders... Urgent Response to Jeong Cheong-rae Terror Plot Reports
- Experts Already Watching Closely..."Target Price Set at 970,000 Won" Only Upward Momentum Remains [Weekend Money]
Spokesperson Ko stated, "The Central Disaster and Safety Countermeasures Headquarters (CDSCH) is preparing prescription and delivery plans for oral antiviral treatments targeting home-treated patients to enable rapid diagnosis and prescription," and added, "Detailed guidance on the introduction, utilization plans, and eligibility for oral antiviral treatments will be provided next week."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.
![[Image source=Yonhap News]](https://cphoto.asiae.co.kr/listimglink/1/2021121510140461697_1639530844.jpg)







