Countdown to Korea's First Domestic Vaccine... Overcoming 'Phase 3 Challenges' to Complete Development
Final Hurdle in Drug Development: Phase 3 Clinical Trials
Costs Exceed 3 Trillion Won
Recruiting Participants and Data Collection Also Challenging
Obstacle Even for Global Pharma Companies
Breakthrough with 'Comparative Trials' Using Existing Vaccine Control Groups

In October last year, President Moon Jae-in visited SK Bioscience in Seongnam, Gyeonggi Province, observing through a microscope in the cell culture room. / Photo by Yonhap News
View original image[Asia Economy Reporter Lim Juhyung] "We must accelerate the development of domestically produced vaccines to avoid being swayed by overseas companies."
This was a remark made by President Moon Jae-in at the Blue House senior secretaries and aides meeting on the 9th. It appears to reflect concerns over the sharp surge in global vaccine demand and production disruption controversies, which have led to difficulties in the smooth supply of vaccines by multinational pharmaceutical companies. At times like these, if a 'domestic vaccine' is supplied, it could greatly contribute to early herd immunity through vaccination. This is why expectations are rising for SK Bioscience's COVID-19 vaccine candidate 'GBP510,' which was the first domestically developed vaccine to receive approval for Phase 3 clinical trials in Korea.
However, for the domestic vaccine to successfully complete clinical trials and enter mass production, it must overcome the hurdle of 'Phase 3.' Recruiting thousands of clinical trial participants and continuously monitoring them is a challenging task even for large multinational pharmaceutical companies.
◆Countdown to the birth of the 'first domestic vaccine'... President Moon promises "comprehensive support"
The Ministry of Food and Drug Safety (MFDS) announced on the 10th that it had approved the Phase 3 clinical trial of GBP510 developed by SK Bioscience. It is the first among vaccines being developed domestically.
GBP510 is a vaccine created using genetic recombination technology, which induces the production of neutralizing antibodies in the human body by injecting the surface antigen protein of the COVID-19 virus. SK Bioscience previously received a total of $213.7 million in funding from the Bill & Melinda Gates Foundation and the Coalition for Epidemic Preparedness Innovations (CEPI) to develop the vaccine, and interim analysis results of Phase 1/2 clinical trials have been released.
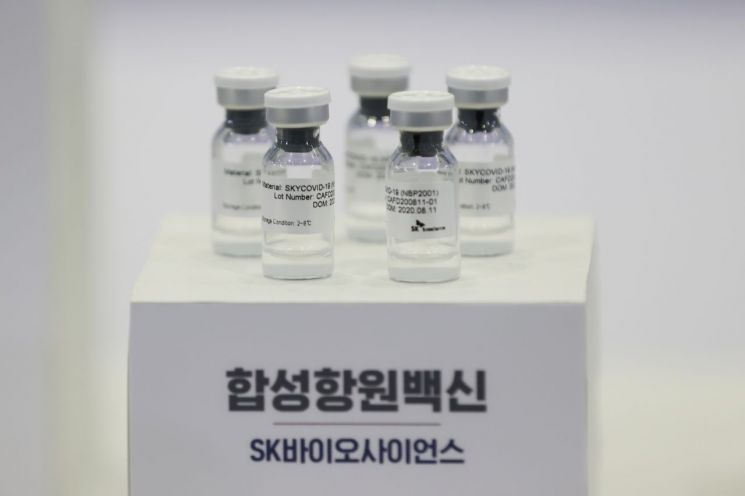
Protein recombinant (synthetic antigen) vaccine reagent of SK Bioscience. / Photo by Yonhap News
View original imageIn Phase 1/2 clinical trials, the level of neutralizing antibody induction by GBP510 was found to be 5 to 8 times higher compared to the serum panel of recovered COVID-19 patients. This is why there is optimism that this vaccine could have significant effectiveness in preventing COVID-19 infection and severe disease.
Regarding this, Blue House spokesperson Park Kyung-mi stated at a briefing on the 10th, "President Moon Jae-in expressed hope that the first domestic vaccine would be born and commercialized, and promised that the government would provide comprehensive support to ensure that domestic clinical trials can be conducted sufficiently and swiftly."
◆Phase 3, the final stage of drug development, is a challenging task even for 'big pharma'
Phase 3 clinical trials are the final stage of drug development and, in the case of vaccines, the most important phase to evaluate protective efficacy and safety against infectious diseases. Therefore, collecting data from as many people as possible is crucial, and typically, more than 1,000 participants are recruited for Phase 3 trials.
Phase 3 trials are known to be the most costly part of drug development. Although costs vary greatly depending on the number of participants and the drug, an essay published in the British scientific journal 'Nature' in 2018 titled "How much does Phase 3 cost?" reported that costs range from a minimum of $43 million (about 49.6 billion KRW) to a maximum of $2.9 billion (about 3.35 trillion KRW).
For COVID-19 vaccines, tens of thousands of participants were recruited for Phase 3 trials. Pfizer had 43,538 participants, Moderna 30,000, and AstraZeneca 23,000. Last year, global pharmaceutical companies administered the real vaccine to some participants and a 'placebo vaccine' to others, closely monitoring their infection rates, severe disease development, and side effects.
Phase 3 trials involve very complex procedures. Participants must be recruited from multiple countries, and specialized research personnel are needed to periodically observe and collect data from them. This is a task only manageable by so-called 'big pharma' companies, which have branches and research institutes in numerous countries and hundreds of researchers. For this reason, BioNTech in Germany and the Jenner Institute at the University of Oxford in the UK had to collaborate with Pfizer and AstraZeneca, respectively, to develop their vaccines.
Large-scale clinical trials are difficult, but another challenge is the difficulty in recruiting participants. This is because widespread vaccination is already underway in many countries worldwide. In developed countries, more than 50% of the population has received COVID-19 vaccines. Additionally, many pharmaceutical companies are conducting vaccine trials, intensifying competition for participant recruitment. This inevitably narrows the position of domestic pharmaceutical companies, which started vaccine development relatively late.
◆Breakthrough through 'comparative clinical trials'
Some pharmaceutical companies have introduced a method called 'comparative clinical trials' to find a breakthrough. Unlike the traditional placebo-controlled method, comparative clinical trials evaluate efficacy by comparing with an already approved vaccine whose safety and efficacy have been established. Because comparative trials require recruiting relatively fewer participants than traditional methods, they can significantly reduce costs and time.
For example, the French pharmaceutical company Valneva is conducting Phase 3 comparative clinical trials in the UK, using the AZ vaccine as the control group. SK Bioscience also plans to conduct Phase 3 trials using the AZ vaccine as the control group in a comparative clinical trial format. The total number of trial participants will be 3,990 adults aged 18 and over.
Hot Picks Today
!["Stock Set to Double: This Company Smiles Every Time a Data Center Is Built [Click e-Stock]"](https://cwcontent.asiae.co.kr/asiaresize/93/2026050416112750184_1777878687.jpg) "Stock Set to Double: This Company Smiles Every...
"Stock Set to Double: This Company Smiles Every...
- "Is Yours Just Gathering Dust at Home? Millennials & Gen Z Rediscover Digicams O...
- "Continuous Groundwater Pumping Causes Mexico City to Sink 24cm Annually... 'Gia...
- "I Take Full Responsibility"... Seongjae Ahn Issues Direct Apology for 'Wine Swi...
- “She Shouted, ‘The Rope Isn’t Tied!’... Chinese Woman Falls from 168m Cliff ...
Regarding this, MFDS Commissioner Kim Gang-rip explained at a briefing on the 10th, "The traditional placebo-controlled method raises ethical issues because participants receiving placebo in a large-scale epidemic are exposed to the risk of infection, and it is practically difficult to secure a large number of participants who have never been vaccinated. As an alternative, we decided to measure vaccine efficacy through comparative clinical trials by vaccinating a small number of participants and confirming immunogenicity."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.








