Samsung Bioepis-Yuhan Corporation, Domestic Sales Partnership for Autoimmune Disease Treatments
[Asia Economy Reporter Seo So-jung] Samsung Bioepis (CEO Go Han-seung) announced on the 15th that it has signed a marketing partnership with Yuhan Corporation for the domestic sales of the autoimmune disease treatment 'Adaloce'.
Adaloce is a biosimilar of the world's top-selling drug 'Humira' and is the first product to receive sales approval in Korea. It is an autoimmune disease treatment for rheumatoid arthritis, ankylosing spondylitis, Crohn's disease, etc., working by inhibiting the expression of tumor necrosis factor (TNF)-alpha in the body.
Since obtaining domestic sales approval for Adaloce in September 2017, Samsung Bioepis has been preparing for its launch through a licensing agreement with the original drug developer AbbVie.
Ahead of the planned launch of Adaloce in the first half of this year, Samsung Bioepis held discussions with multiple companies to select a marketing partner and chose Yuhan Corporation, which has high expertise in the domestic prescription drug market, as the sales company.
With this, Samsung Bioepis will sell biosimilar products for all three blockbuster domestic TNF-alpha inhibitor markets (Humira, Enbrel, Remicade), which are worth approximately 200 billion KRW.
Samsung Bioepis is also conducting domestic sales of its previously launched autoimmune disease treatments 'Etoloce' (Enbrel biosimilar) and 'Remaroce' (Remicade biosimilar) through Yuhan Corporation.
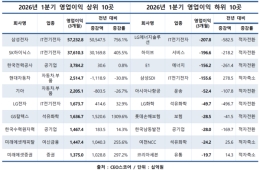
Hot Picks Today
 "Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
"Now Our Salaries Are 10 Million Won a Month" Record High... Semiconductor Boom Drives Performance Bonuses at Major Electronic Component Firms
- Experts Already Watching Closely..."Target Price Set at 970,000 Won" Only Upward Momentum Remains [Weekend Money]
- "Heading for 2 Million Won": The Company the Securities Industry Says Not to Doubt [Weekend Money]
- 'Strait of Hormuz Blockade' Prompts 57 Countries to Intervene in Markets... Oil Price Caps and Fuel Tax Cuts in Full Force
- Is It Really Like an Illness? "I Can't Wait to Go Again"—Over 1 Million Visited in Q1, Now 'Busanbyeong' Takes Hold [K-Holic]
Go Han-seung, CEO of Samsung Bioepis, said, "We are pleased to be able to introduce biosimilar products of the world's most widely used drugs in Korea, and we will continue to strive to expand treatment opportunities for patients through high-quality medicines."
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.